Good news on vaccines vs variants!
Tagged:COVID
/
JournalClub
/
MathInTheNews
/
PharmaAndBiotech
/
Statistics
Some good news, for once: the Pfizer/BioNTech vaccine is now known to work quite well against the British & South African variants in the real world. Especially in preventing hospitalization and death.
What’s the Sitch?
This exceptionally annoying virus is continuing to mutate. These were the high points of the mutation situation last February [1], and since then it’s only gotten worse, with more variants:
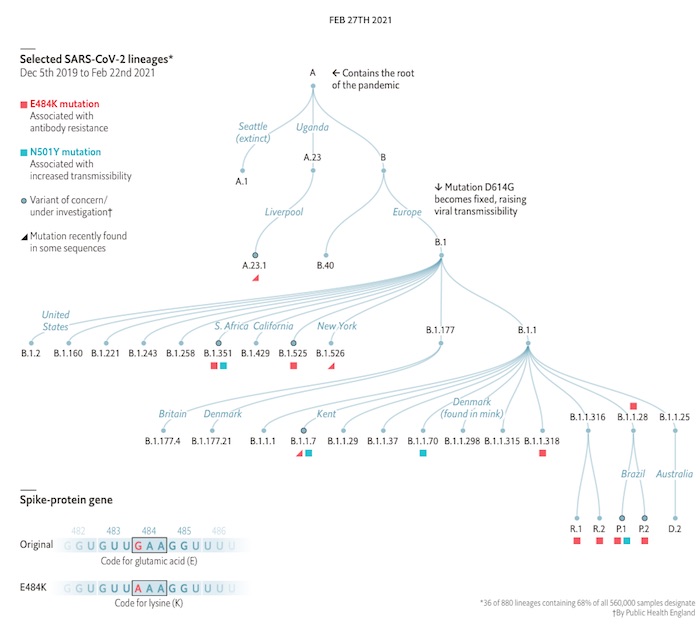
There are 3 things to worry about with a new variant:
- Does it spread faster? Increased affinity of the spike protein’s receptor binding domain (RBD) for human ACE2 protein will cause this. More cases, more deaths.
- Is it more deadly? A higher replication efficiency will do this. Same number of cases, but more deaths.
- Is it an escape mutation, i.e., evading vaccine protection? The vaccines are trained on the spike protein of “corona classic” (with some very clever modifications we wrote about previously). A modified spike protein could get around that.
So far, we’ve seen pretty good evidence of worry #1, higher spread, in both the B.1.1.7 (British, or as a British friend puts it “the Kent strain” for the county where it was found) and B.1.351 (South African) variants.
Fortunately, there doesn’t yet seem to be strong evidence of #2, higher mortality.
But what about worry #3, vaccine escape mutations? The evidence so far has been
equivocal, and largely in vitro:
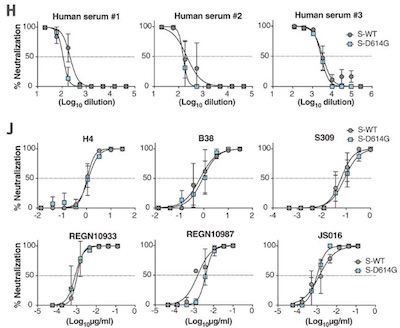
- Last December, we looked at a paper in Science by Hou, et al.[2] showing that antibodies still had good neutralization curves against a D614G variant. That’s nice; a beautiful bit of work, even. But it’s not in people, or even in an animal, and it’s not one of the exact variant’s that are so scary. So it’s a glimmer of hope, but just a glimmer.
- Then in March, we looked at some news analysis by Wise in the BMJ [3] which was far less hopeful. Apparently the required antibody concentration for neutralization of the E484K variant was much higher. Ruh roh…
- Fortunately, in that same post we also saw some evidence from Redd of NIAID [4] that CD8+ T cells were still active against the British, South African, and Brazilian variants. So even if it takes more antibodies, your immune system remembers and knows it can kill cells infected with variants.
Today’s Good News
So… mixed evidence, all of it in the lab and not in people or even animals.
 That’s why I was very happy to see a news report in the
New York Times [5] alleging they had evidence that the
Pfizer/BioNTech vaccine worked to prevent the most severe outcomes (hospitalization &
death: remember, the goal is not to die) in some SARS-CoV-2 variants. The summary:
That’s why I was very happy to see a news report in the
New York Times [5] alleging they had evidence that the
Pfizer/BioNTech vaccine worked to prevent the most severe outcomes (hospitalization &
death: remember, the goal is not to die) in some SARS-CoV-2 variants. The summary:
- These 2 studies were done on large, real-world popuations in Qatar and Israel. So it’s not in vitro lab data, but actual human beings going about their lives, misbehaving in various virus-sensitive ways, and so on. The real world.
- Sequencing indicated most of the infections in those countries were variants of concern: B.1.1.7 and B.1.351.
- While the AZ/OX vaccine appears to be somewhat less effective against B.1.351, Pfizer/BioNTech had good efficacy against infection and near-perfect efficacy at preventing severe disease and death.

 While the NYT has its problems with rampant both-siderism, this looked worth digging
into. So I had a look at the papers: the Qatari study by Abu-Raddad, et al. is pubished
in the NEJM [6], and the Israeli study by Haas, et al. is
published in The Lancet. [7]
While the NYT has its problems with rampant both-siderism, this looked worth digging
into. So I had a look at the papers: the Qatari study by Abu-Raddad, et al. is pubished
in the NEJM [6], and the Israeli study by Haas, et al. is
published in The Lancet. [7]
Each of them follows real-world populations, country-wide. This leads to very good statistics indeed: 265,410 Qataris had both doses and more than 230,000 Israeli cases. (That’s the sort of good stuff that happens when you have a well-integrated national healthcare system with good electronic medical records. Maybe one day the US will do that, and rejoin the developed world.)
Each of them looks, at first glance, to be statistically sound: they did near-universal sampling of their national healthcare system to avoid bias, they calculated 95% confidence limits on their efficacies, and all that. (We’ve looked at how to calculate vaccine efficacy confidence limits before, so this is checkable.) While I haven’t looked through their methodologies with a fine-tooth comb, it looks great to me and they’re publishing in extremely high quality academic journals anyway.
So if we decided to believe them, what would we end up believing?
Here’s the summary table from the Qatari study:
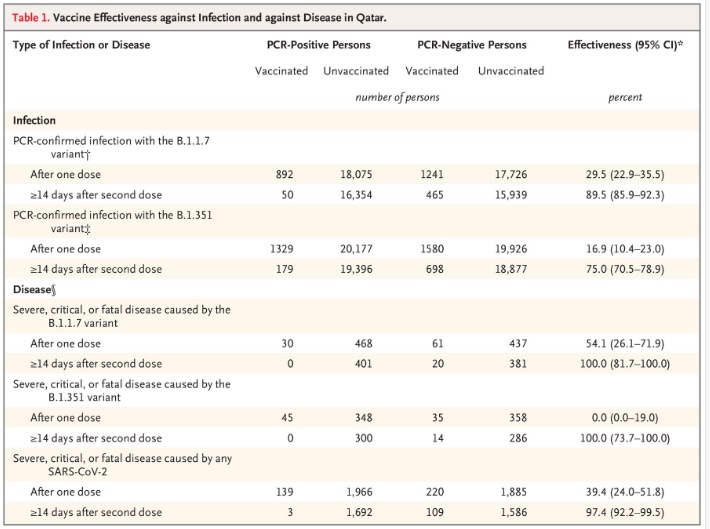
- It first looks at efficacy against any COVID-19 infection at all. It looks like, 14 days after the second dose of Pfizer, the efficacy is 89.5% (CL: 85.9% - 92.3%) against the British variant, and 75.0% (CL: 70.5% - 78.9%) against the South African. (Check out those beautifully narrow confidence limits! Large sample size, my dudes. Large samples size.)
- That’s less than the 95% from the original clinical trial, but recall that annual flu vaccines save lives with 50% - 60% efficacy. This is way more than enough!
- If you restrict attention to severe, critical, or fatal disease (defined in the paper), then the efficacy is 100% against both variants (with wider confidence limits, as appropriate for something that almost never happens).
- Best of all: the efficacy against severe disease from any SARS-CoV2 at all is 97.4% (CL: 92.2% - 99.5%). That’s spectacular.
Here’s the similar result table from the Israeli study:
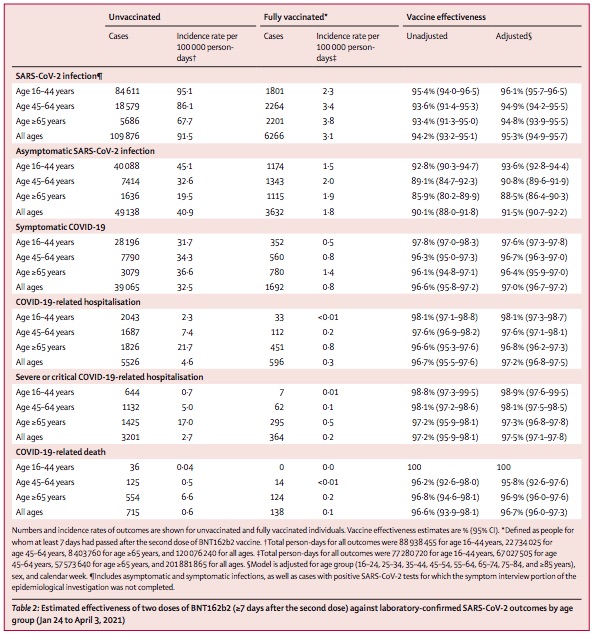
- First, we note that they are talking about a population infected by B.1.1.7 in almost 95% of the cases. Fast-spreading variants crowd out all the others.
- Note also that they break down their results by age group and by severity (any infection, asymptomatic, symptomatic, hospitalized, critical, and deadly). So you can look at all sorts of strata, depending on your interest. We should be most interested in hospitalization or worse, because: the goal is not to die. (I dunno why I have to keep repeating that, but here we are.)
- I like this table: it makes it easy to answer questions that are of great concern to me. For example, as mid-60s sort of people who are 2 weeks past our 2nd dose of Pfizer, what’s the efficacy of our vaccine against COVID-19 death due to B.1.1.7? See the last row of the table: 96.7% (confidence limit: 96.0% - 97.3%). Very reassuring. (Though, maybe we have to emigrate to Israel to make this apply to us? Hmmm.)
Absolutely fabulous result! Both scientifically (big, clean datasets analyzed clearly) and in in terms of the good news they bring. This, dear readers, is how it is done.
The Weekend Conclusion
In particular, the table above from the Abu-Raddad study is hopeful: if you can get to 14 days past the second dose of Pfizer/BioNTech (though really, Moderna is likely to be nearly identical), then your risk is virtually eliminated for severe/critical/fatal disease with SARS-CoV2 classic, British, or South African variants.
The table from the Haas study is also chock-full of good news for us here at Chez Weekend, as sixty-somethings: the efficacy vs severe disease from the British variants is 96% - 97% in our age group.
I’m relieved to note that both your humble Weekend Editor and Weekend Editrix are now fortunate enough to find ourselves in that position (as we hope all humanity can be soon). Maybe we can venture forth out of our front yard sometime?
We don’t know yet about the Indian or Brazilian variants, and we don’t know about the JnJ or AZ/OX vaccines. But at least one thing is working well.
Addendum 2021-May-09: Moderna, too. Also, P.1 from Brazil.


Moderna issued a press release [8] and an (not yet peer reviewed) paper [9] on the medR$\chi$iv preprint server.
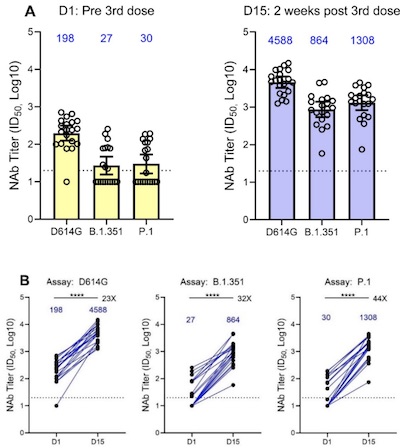
- This is a preliminary readout from a Phase 2 trial of 3 different strategies:
- Just a 3rd booster shot of mRNA-1273 (the Moderna vaccine now in use)
- A booster called mRNA-1273.351, which is tailored to the South African strain’s spike protien
- A combined booster mixing both of those 1:1, to be called mRNA-1273.211
- They measured neutralizing antibody titers with a pseudovirus neutralization assay, using classic SARS-CoV-2, the South African variant (B.1.351) and the Brazilian variant (P.1). Response was good in all cases: 12x - 35x higher, if I’m reading their figure right.
- The statistics were nice and straightforward: geometric mean titer on log-tansformed titers, with a 95% CI calcluated from a $t$-distribution and significance from a Wilcoxon matched-pairs signed rank test.
- The side effects were comparable to the previous doses.
This, combined with similar news from Pfizer and a promise from the FDA of rapid review (comparable to the review done on an annual flu vaccine), makes for very good news.
Notes & References
1: Public Health England graphic, “The same covid-19 mutations are appearing in different places”, The Economist, 2021-Feb-27. ↩
2: Y Hou, et al., “SARS-CoV-2 D614G variant exhibits efficient replication ex vivo and transmission in vivo”, Science 370:6253 (2020-Dec-18), pp 1464-1468. DOI: 10.1126/science.abe8499 ↩
3: J Wise, “Covid-19: The E484K mutation and the risks it poses”, BMJ 2021, 372:n359. ↩
4: A Redd, et al., “CD8+ T cell responses in COVID-19 convalescent individuals target conserved epitopes from multiple prominent SARS-CoV-2 circulating variants”, Open Forum Infectious Diseases ofab143, 2021-Mar-30. DOI: 10.1093/ofid/ofab143.↩
5: E Anthes, “Covid-19: Studies Indicate Pfizer-BioNTech Vaccine Protects Against the Most Severe Outcomes of Some Virus Variants”, New York Times live blog, 2021-May-06.↩
6: L Abu-Raddad & A Butt, “Effectiveness of the BNT162b2 Covid-19 Vaccine against the B.1.1.7 and B.1.351 Variants”, Letters to The New England Journal of Medicine, 2021-May-05. DOI: 10.1056/NEJMc2104974.↩
7: EJ Haas, et al., “Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: an observational study using national surveillance data”, The Lancet (in press, corrected proof) 397:10285, 2021-May-1-7. DOI:10.1016/S0140-6736(21)00947-8. ↩
8: C Hussey (media contact), “Moderna Announces Positive Initial Booster Data Against SARS-CoV-2 Variants of Concern”, Moderna Press Releases, 2021-May-05.↩
9: K Wu, et al., “Preliminary Analysis of Safety and Immunogenicity of a SARS-CoV-2 Variant Vaccine Booster”, medR$\chi$iv, 2021-May-06. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.