FDA VRBPAC: Omicron-Specific COVID-19 Boosters?
Tagged:COVID
/
MathInTheNews
/
PharmaAndBiotech
/
Politics
/
Statistics
Today the FDA’s Vaccine and Related Biological Products Advisory Committee (VRBPAC) met to consider whether to add Omicron-specific vaccines to the mix, and how that policy should be set. Wanna read along to see what they do?
Omicron-specific boosters (preview: the day before the VRBPAC meeting)
Ahead of the meeting, only the meeting agenda [1], meeting roster [2], discussion questions [3], voting question [4], and the FDA analysis [5] have been published so far. (And a couple disclosures of financial interest and the associated waivers, which do not concern us here.)
The real content will be in the slide decks and the industry submissions, which I expect will start appearing either late tonight or early tomorrow morning.
In the meantime, that means the FDA analysis document is the most important scientific thing publicly visible so far. So let’s have a preview look at that.
There’s lots of stuff there, but the first impression I have is brevity: it’s only 22 pages, including title, contents, and references. For these folk, that’s the soul of brevity (if not wit).

 There are some interesting sections outlining the possible policies
the VRBPAC can choose, but the most interesting is the efficacy data submitted by Pfizer
and Moderna. Moderna previously released their topline summary in a press
release [6], about which
we blogged previously. Pfizer issued a similar
press release just a couple days ago [7] (which we didn’t
blog separately, because this meeting was coming up in just 3 days).
There are some interesting sections outlining the possible policies
the VRBPAC can choose, but the most interesting is the efficacy data submitted by Pfizer
and Moderna. Moderna previously released their topline summary in a press
release [6], about which
we blogged previously. Pfizer issued a similar
press release just a couple days ago [7] (which we didn’t
blog separately, because this meeting was coming up in just 3 days).
The FDA summary is of both trial results is in §3.5, pp. 13-15. The 2 criteria are:
- Non-inferiority on previous strains: Don’t mess up immune response to previous strains. I.e., the antibody levels should be at least as good as the existing vaccine on previous strains because the last thing we need is Delta revenant!
- Superiority on Omicron strains: Do better than the previous vaccines on Omicron strains, exact strains left up to what was in circulation at the time of the clinical trial.
The trials compared adults who’d gotten 3 shots of the previous vaccines, and had no
evidence of prior infection. The control arm got a 4th shot of the same, while the
treatment arm got a 4th shot of an Omicron-specific mRNA vaccine. The FDA doc doesn’t
summarize the results in a table, instead throwing up a maddening barrage of word salad.
(Let’s hope the presentations tomorrow are more straightforward!) But after hacking my
way through it, the main results seem to be what’s shown in the table here, that we built
from data extracted from the word salad:
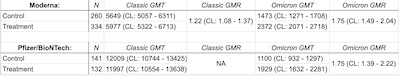
- GMT is the geometric mean titer, i.e., antibody levels. GMR is the geometric mean ratio, i.e., Omicron/Classic vaccines where a ratio > 1 is favorable.
- Numbers in parentheses are the 95% confidence limits.
- The Pfizer trial was about half the size of the moderna trial.
- Against the classic viral strains:
- Both the Omicron-specific vaccines were pretty comparable to the classic vaccines when confronted with the classic viral strains. Pfizer didn’t compute a GMR and its confidence limits, but clearly the numbers are comparable.
- The GMT numbers for Pfizer are dramatically higher than Moderna, and I don’t know why. I hope the presentations will bring clarity about the units used and the absolute scale.
- Against the Omicron viral strain:
- Both Omicron-specific vaccines did better, with a GMR of about 1.75 and bounded above 1.0 in the 95% confidence limit.
- Unlike the classic virus, here the Pfizer GMT numbers are comparable to the Moderna numbers. Again, I don’t quite know why.
- In both cases, the GMT’s are lower for Omicron compared to the classic virus, but the Omicron-specific vaccine is still better. Omicron is a tough target!
- Pfizer also did a study of patients over age 55, at both 30μg and 60μg doses, reaching similar conclusions, but I won’t get into the weeds on that until the presentations tomorrow.
So to me, that looks like a very good case for rolling out Omicron-specific vaccines. (How exactly that will be done is another question, e.g., whether to just replace the old vaccines altogether.)
It seems the FDA analysts agree with us:
The data from these two studies suggest that an Omicron monovalent vaccine or Omicron plus ancestral bivalent vaccine as the 2nd booster vaccination (4th dose) improves the neutralizing antibody response to Omicron BA.1 compared to the prototype vaccine and does not negatively affect the neutralizing antibody response against the ancestral strain of virus against which the prototype vaccine was designed.
Let’s see if the VRBPAC agrees. Or, more realistically, what they want to argue about.
The discussion questions and voting questions
The discussion questions are:
- Should we change our vaccines from the classic ones at all?
- If so:
- Which Omicron sub-lineages (BA.1 vs BA.2 vs BA.4/5 vs …) should be used?
- Should it be monovalent (Omicron alone) or bivalent (Omicron + classic)?
- Is it any different for different age ranges?
- What else should we know?
Interestingly, it’s far too late to change the composition of the vaccines now, and expect production by fall. We can’t even turn around a clinical trial in the remaining time. So I wonder where the “discussion” will go, and to what point.
The voting question is simpler:
Does the committee recommend inclusion of a SARS-CoV-2 Omicron component for COVID-19 booster vaccines in the United States?
Not a word about strain composition, mono- vs bi- valent, whether to keep using the classic vaccines as primaries with Omicron boosters vs all Omicron all the time, etc.
It should be interesting to hear the arguing about that. It’s important stuff, and I wish we’d gotten our act together a lot faster to decide the optimal strain and vaccine administration pipeline. As it is, we either stick with what we’ve got or take what’s on offer from the vaccine makers.
The FDA overview
After the usual preliminaries, introductions, statements of conflicts of interest, roll calls, and so on… was Peter Marks of the FDA. [8] Referring to past guidance from the FDA on EUA’s for COVID-19 boosters [9], he made basically 3 points as shown here in 3 slides:
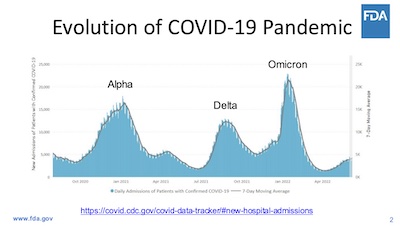
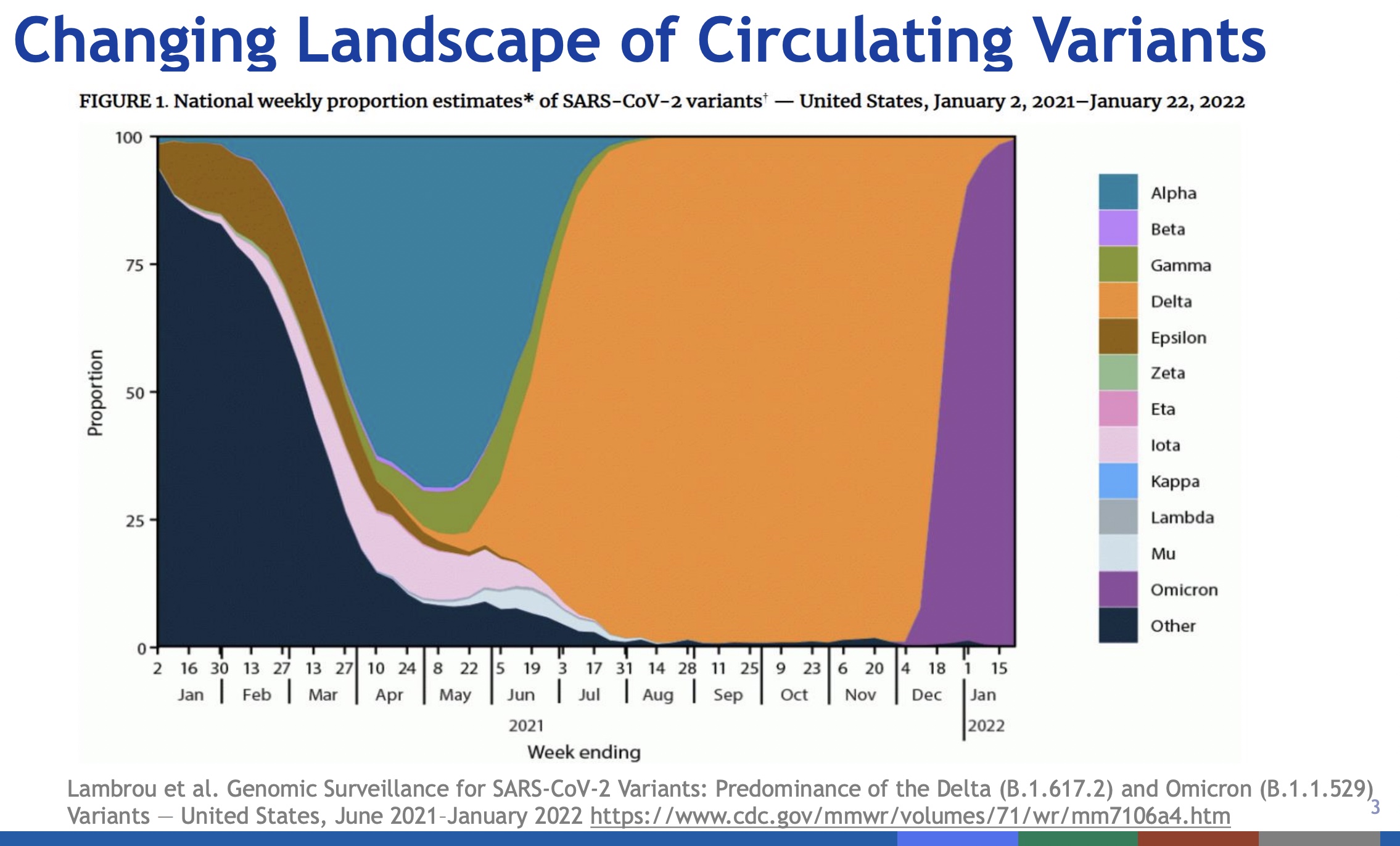
- Slide 2: The pandemic is not over, regardless of how people feel. The wave structure of the pandemic over time is clear, as is the current rise in hospitalizations. Within weeks, the COVID-19 cases in the US will be 100% Omicron BA.5 variant (vs 20% today).
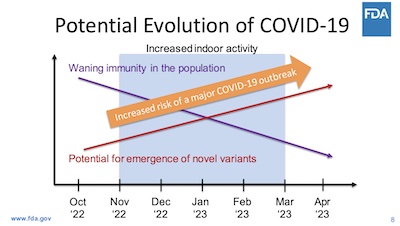
- Slide 8: Vaccines help. This slide is of course way over-simplified, but it has that kick-in-the-gut simplicity necessary for communications with a poorly informed public. As immunity wanes and the virus evolves, the harm from a new wave increases.
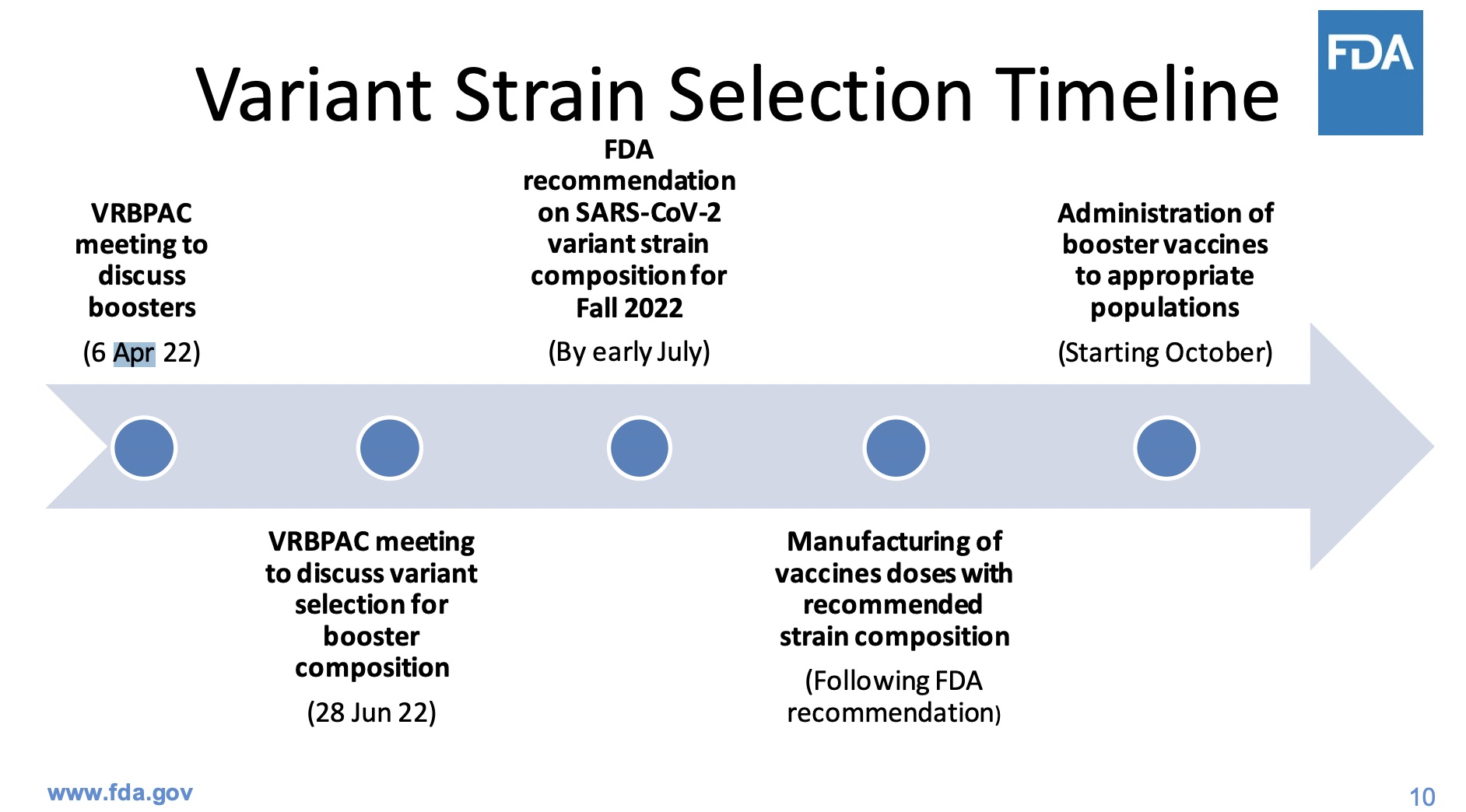
- Slide 10: He has an unrealistically optimistic view of the timeline for variant-specific vaccines, apparently regarding manufacturing as a little technical detail that can be ignored. This is where I reluctantly part company with him: he thinks the FDA can specify the strains to go into a vaccine in early July and have testing & manufacturing go fast enough for administration by October. That’s… kinda nuts.
Still, let’s give him credit for having his heart in the right place. He just needs a little more industry guidance on testing, manufacturing, and distribution time and capital requirements.
The CDC overview
Next came some updates from the usual suspects at the CDC, Heather Scobie on SARS-CoV2 epidemiology[10] and Ruth Link-Gelles vaccine efficacy against Omicron variants. [11]
Scobie said a lot of things, but here are the high points:
- Slide 3: Omicron has taken over, and fast. It’s all that matters now. Soon it will all be the BA.5 subvariant of Omicron. The strong structure shown here of waves of variants overcoming the previous variants is a portrait of a fast-evolving virus. It’s evolving to feed on us.
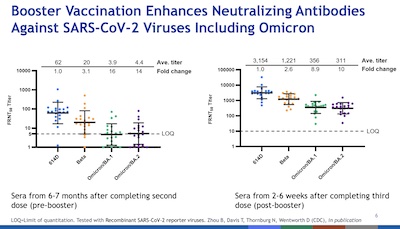
- Slide 6: Immunity wanes over time, as the virus evolves. However, boosters help remarkably well. That’s mostly a brute-force effect, i.e., hitting the newer variants with a wall of antibodies. There’s some antibody maturation in the immune system, of course, but mostly this is just brute force and dumb luck that the old vaccines continue to work against new variants. Note the log vertical scale: boosters raise titers by 2-3 orders of magnitude!
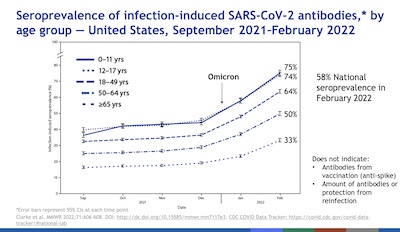
- Slides 15 & 16: There’s been a lot of interest in figuring out how many people
have already been infected, whether they know it or not. That’s the object of the
nucleocapsid antibody assays we recently wrote about.
Scobie shows 2 interesting results:
- It’s mostly the young who get infected. Their elders are either more vaccinated, more cautious, or both.
- On the other hand, when you look at hospitalizations, it’s reversed: mostly the elderly suffer.
- Conclusion: Young folk may think they’re invulnerable, and indeed they face a lower risk of hospitalization and death. But they can act as carriers to infect and kill their older family and friends. Maybe don’t be a carrier? Getting vaccinated even if you’re young is a good way to do that!
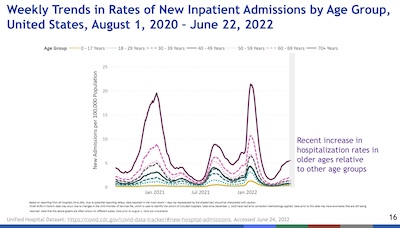
- Slides 24 & 28: These show another interesting age-related dichotomy.
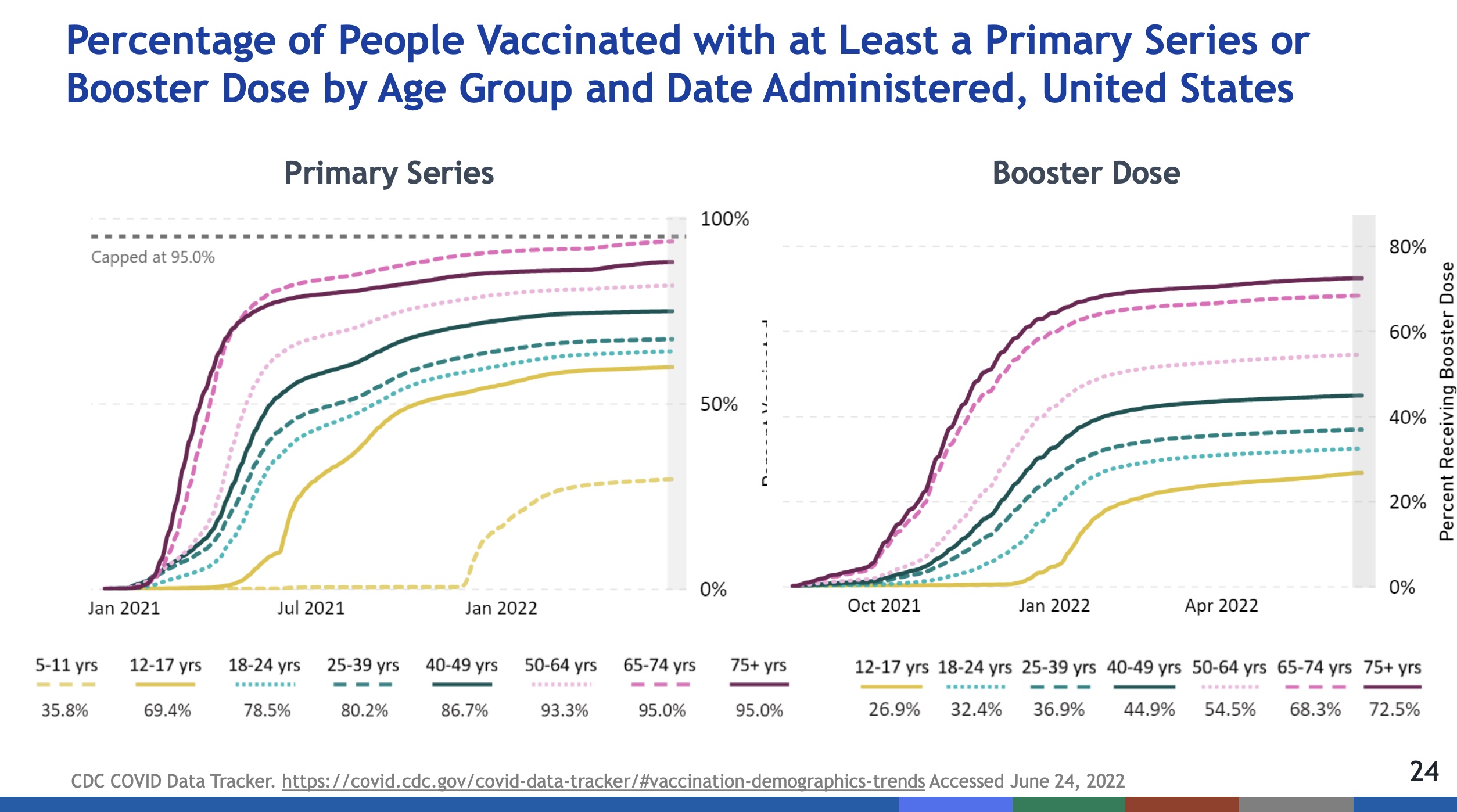
- On the one hand, vaccination & booster uptake over time clearly stratifies by age. The oldest clades are the most vaccinated, and the youngest the least (leaving aside the only recently-approved vaccinations for the under 5 crowd). Again, perhaps the middle-aged and younger feel they face no threat, but they can certainly be asymptomatic carriers.
- On the other hand, hospitalization & death rates over time show almost all the harm is being done to the unvaccinated. The vaccinated but not boosted suffered a little, and the vaccinated & boosted suffered least.
- Conclusion: You may think you’re personally at low risk, but (a) you’re placing older people around you at risk and (b) taking on most of the pandemic’s death rate yourself. Getting vaccinated, regardless of age, seems like an excellent idea.
Link-Gelles had some very disturbing results on vaccine efficacy waning, and some good news about the effect of boosters. There are lots ways to slice-n-dice the data (by age cohort, by vaccination level, by days since last shot, by disease severity/hospitalization/death, etc.).
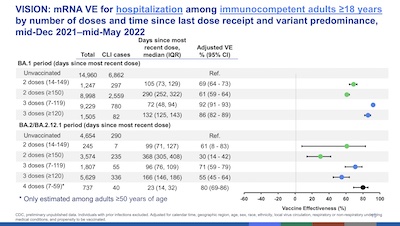 She does a lot of them! We’ll take the advice of our safari guides Helen & Matt at STAT
News here, and just show this summary slide. It shows vaccine efficacy vs
hospitalization. It’s broken down Omicron wave (BA.1 vs BA.2), by number of doses, and by
days since last dose.
She does a lot of them! We’ll take the advice of our safari guides Helen & Matt at STAT
News here, and just show this summary slide. It shows vaccine efficacy vs
hospitalization. It’s broken down Omicron wave (BA.1 vs BA.2), by number of doses, and by
days since last dose.
Conclusions:
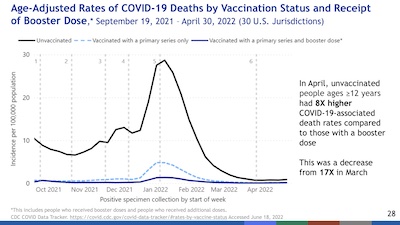
- Efficacy is lower for Omicron than previous variants (hence the need for Omicron-specific boosters).
- Efficacy fades on a timescale of 3-6 months since the last booster.
- There is considerable uncertainty (error bars) in the case of BA.2/BA.2.12.1.
- Boosters, even the now-classic one, help restore hospitalization efficacy to about 80%.
So that’s a mix of bad news and good news: efficacy fades and the virus is evolving ways to evade it, but boosters still work well enough. It’s an important question how long they will continue to work, and whether an Omicron-containing booster is worhtwhile.
What viral surprises might the future hold?
That exact question (how things might change in the future) was the subject of the next presentation, by Justin Lessler of the COVID-19 Scenario Modeling Hub (affiliated with a dozen or so academic and medical institutions). [12]
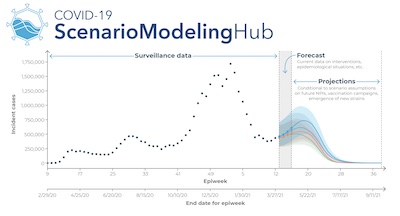 They take combined outputs of an ensemble of 10 models, attempting to get consensus
predictions. They think of their model as dividing the pandemic into 3 phases: the
surveillance data from the past, a short-term forecast based on current policies, and a
longer-term prediction (usually with wide error bars, as shown here in an overly optimistic
sample from last year).
They take combined outputs of an ensemble of 10 models, attempting to get consensus
predictions. They think of their model as dividing the pandemic into 3 phases: the
surveillance data from the past, a short-term forecast based on current policies, and a
longer-term prediction (usually with wide error bars, as shown here in an overly optimistic
sample from last year).
In this round of scenario building, they consider 4 cases:
- Either waning of protection vs infections is viewed optimistically (10 months lose 40%
of protection) or pessimistically (faster waning in 4 months down to 60% efficacy).
- Link-Gelles’s data above indicates the pessimistic version is more realistic.
- Either no new variant by 2022-May-01 or there is one with a certain rate of new
hospitalizations for 16 weeks, 30% immune escape, and about the same $R_0$ and
severity.
- The observation of multiple Omicron variants favors the latter, darker, assumption.
A new variant will lead to earlier surges, and faster waning will lead to higher hospitalization rates.
Here are a couple of their results:
- Each slide shown here has 4 different cases, corresponding to the 2
pessimistic/optimistic assumptions above. The doubly pessimistic one, which we think
most reasonable, is in the lower left.
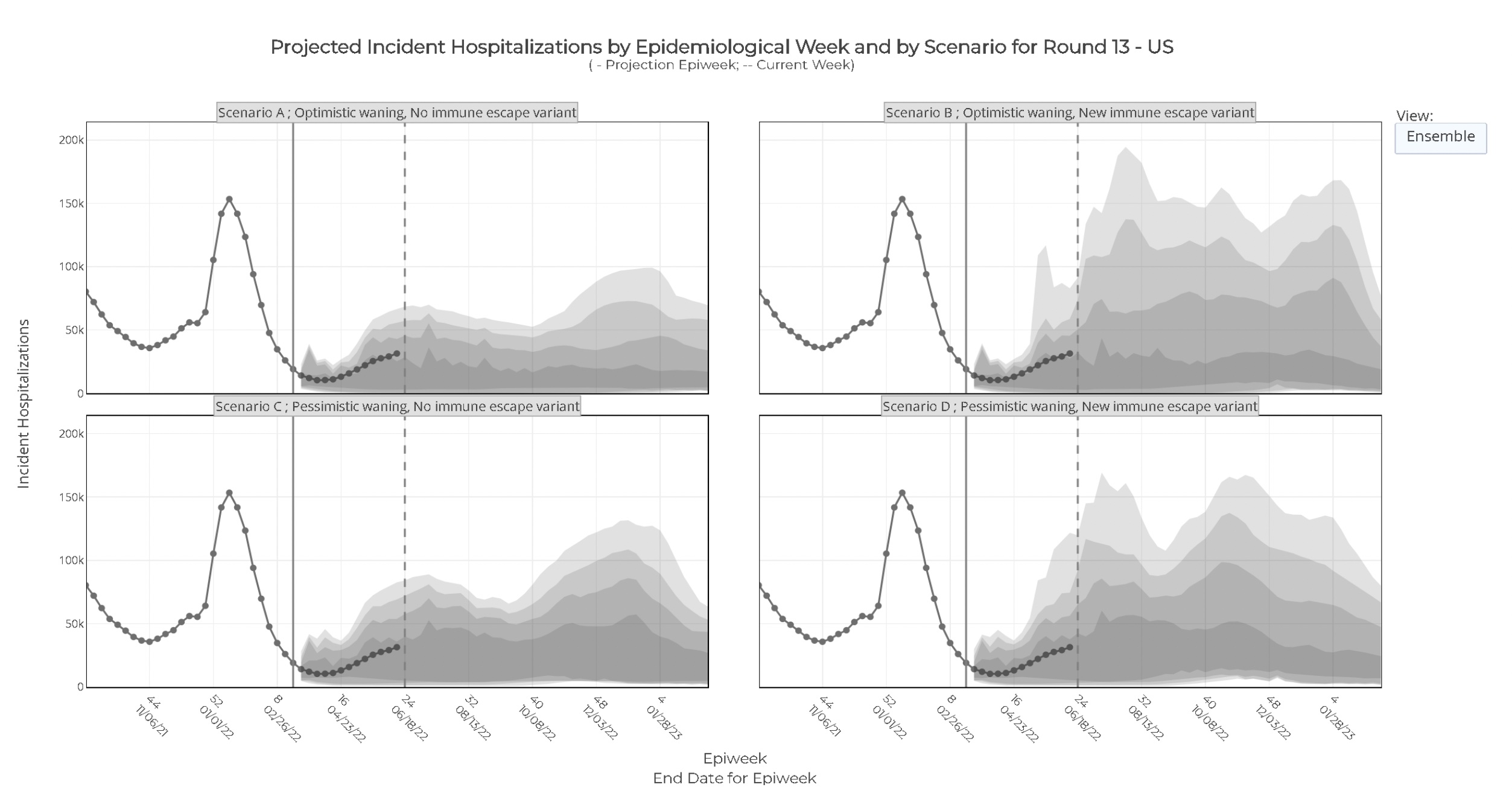
- The top slide shows the time course of hospitalizations as predicted by the ensemble
of models.
- The color bands indicate the 50%, 80%, 90%, and 95% confidence limits, respectively.
- They all predict a winter surge, with the double-pessimistic version being the worst.
- There is however, a considerable band of uncertainty. This is how you recognize good
modeling work: you’re given not just a number, but a distribution of numbers with
probabilities assigned.
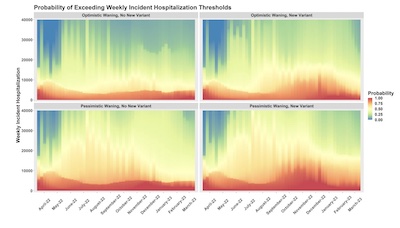
- The bottom slide, in color, shows what amounts to the cumulative distribution function
of hospitalization predictions at each time.
- Red means there is nearly 100% probability that hospitalizations will be above that level at that time. This is the best-case scenario the ensemble can make for low hospitalizations, and you must plan for at least this much hospitalization!
- Blue means there’s almost 0% probability of hospitalizations being above that level at that time. This is the worst-case scenario the ensemble can make for high hospitalizations, and we need not plan on levels above this.
- The doubly pessimistic version predicts the worst of hospitalizations to peak in this fall through December.
The most pessimistic scenario, which they seem to agree is the most realistic, predicts hospitalizations of 13k - 52k per week, likely remaining under 170k per week. There’s a 5% chance of exceeding the worst hospitalizations of Delta.
In that same pessimistic scenario, they project a cumulative number of deaths from 2022-Mar to 2023-Mar of 211,000 (95% CL: 52,000 - 466,000) deaths.
Those are not fun numbers.
Sponsor presentations: Moderna, Pfizer, and Novavax
Now on to the main event: how did the clinical trials of variant-specific, sometimes bivalent, vaccines work out?
Moderna
Moderna’s entry [13] is euphoniously yclept “mRNA-1273.214” after its famous parent “mRNA-1273”. It is a bivalent vaccine, containing mRNA for the spike in the classic Wuhan variant as well as Omicron. The specific Omicron variant is B.1.1.529, which is ancestral to the current pests BA.1 through BA.5. (The B.1.1.529 was the only Omicron strain in circulation at the start of the trial.) Both are dosed at the 25μg level, so that’s a total of 50μg matching the existing boosters.
It’s worth noting that Moderna has previously done a trial with a bivalent vaccine of ancestral Wuhan + Beta variant (B1.351). That worked, but the virus moved on before we could get the trial done, submission to the FDA, approval, and manufacturing. So mRNA-1273.211 is kind of a “zombie” vaccine for a combination no longer relevant.
Let’s hope we move faster with this one.
They had to show 3 things to get the FDA to consider approval:
- Superior geometric mean titers (GMT, i.e., antibody levels) against Omicron.
- Non-inferiority of seroresponse rate (SRR) against Omicron.
- Non-inferiority of GMT & SRR against ancestral variant.
Basically: make lots of Omicron abs, make your immune system capable of responding to Omicron, and don’t mess up immunity to previous strains.
They achieved all of those. Also, reactogenicity (how crappy you feel for a day or two) was actually slightly less of a problem than their original vaccine.
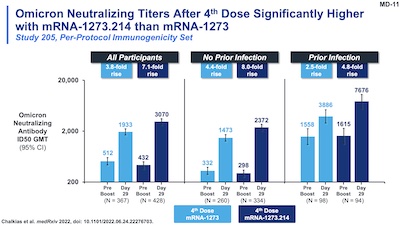
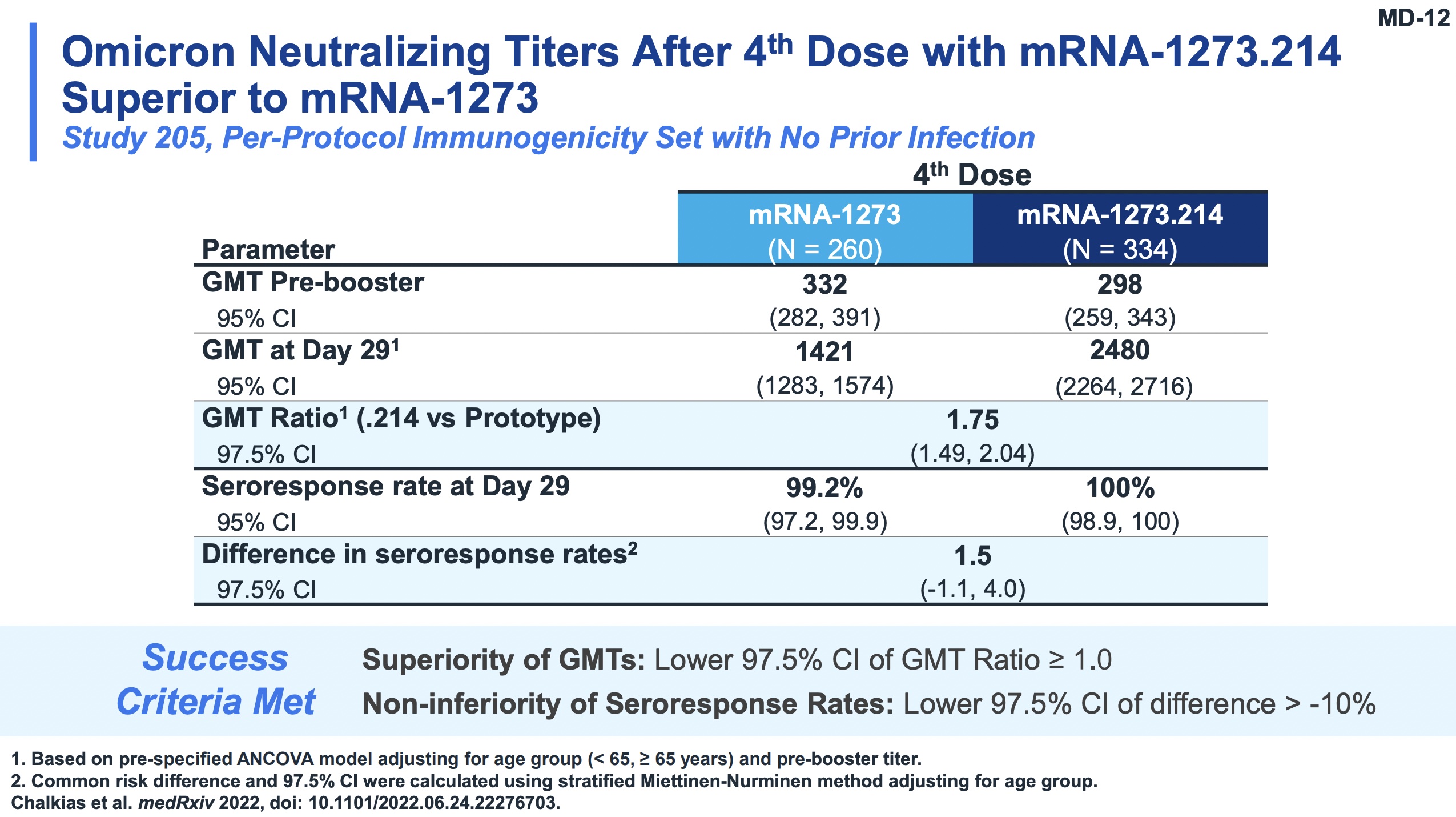
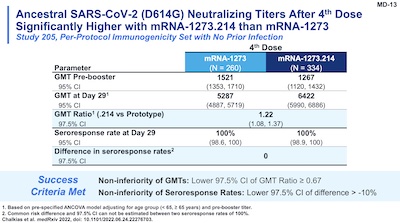
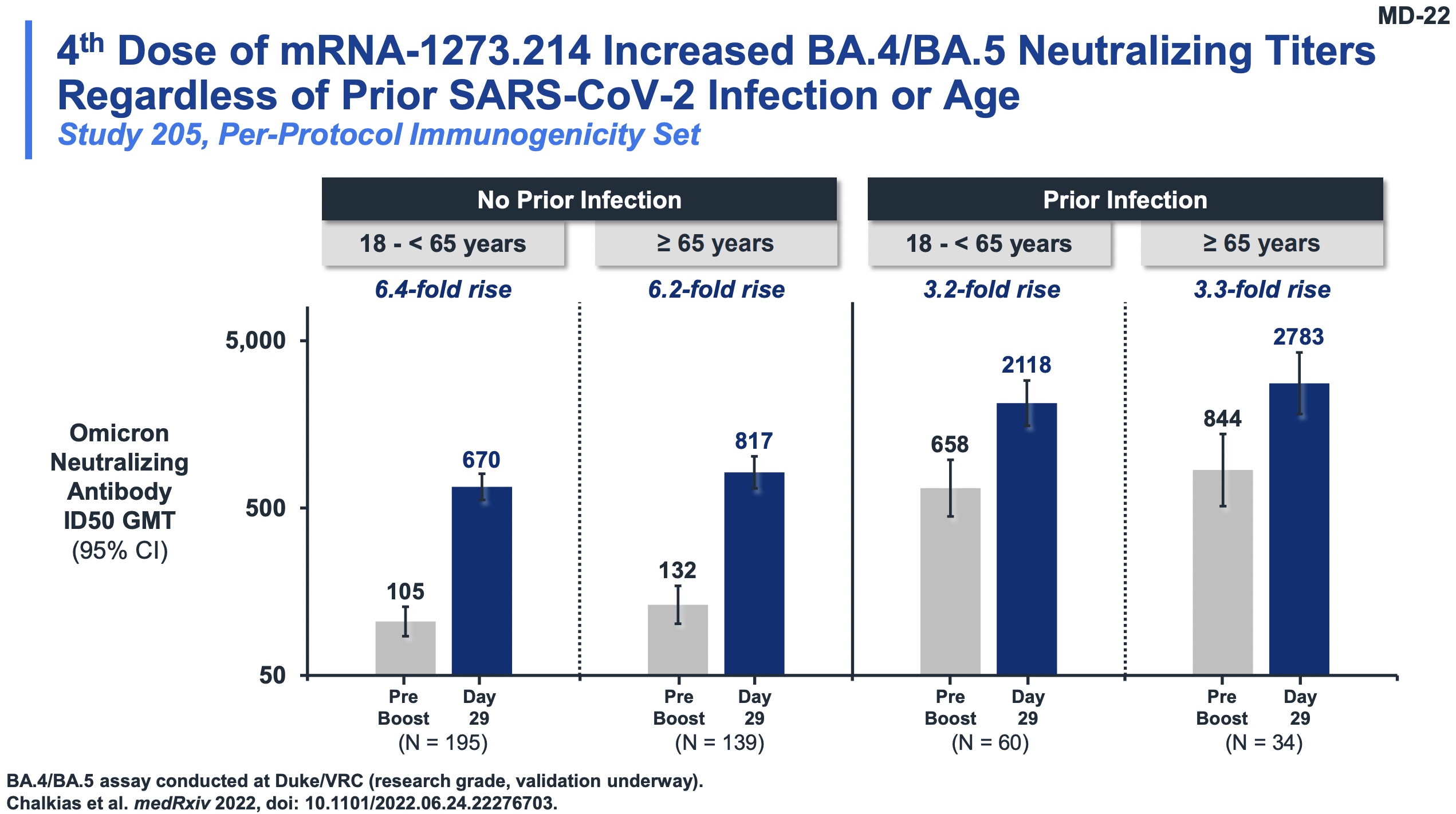 Slides 11-13 and 22 of their presentation make this point dramatically.
Slides 11-13 and 22 of their presentation make this point dramatically.
- Slide 11: Looking at the Omicron neutralizing ab ID50 GMTs, comparing the classic vaccine vs the bivalent Omicron one, we see higher fold induction with the new vaccine than the old one.
- Slide 12: To compare those quantitatively, we see the ratio GMR between the old and new vaccines on Omicron samples was 1.75 (95% CL: 1.49 - 2.04). Also looking at seroresponse rates, we see non-inferiority (it’s hard to improve much from 99.2%, but the bivalent vaccine did so at 100%).
- Slide 13: Doing the same comparison of vaccines but now on the ancestral strain, we see a similar result. The GMR ~ 1.22 (95% CL: 1.08 - 1.37) and SRR non-inferiority (100% in both cases).
- Slide 22: These results are robust across age groups and previous infection status.
- Other slides (I won’t drag you through everything!) show that this did not depend on age, SARS-CoV2 variant, or many other variables. Also, the levels of antibodies induced are clinically significant, matching the level of antibodies that fought off the Delta variant.
This is what success looks like!
Pfizer
Next in our Canterbury Tales of vaccines was a presentation from Pfizer. [14]
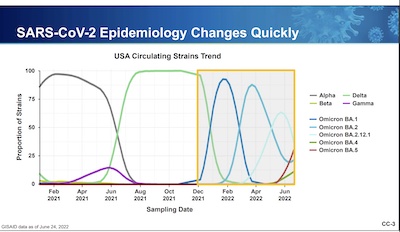 They begin with an excellent summary of how dire the situation is:
They begin with an excellent summary of how dire the situation is:
- It used to take a decade or so to make a vaccine. Ok, we’re better at that now, but…
- 2021 alone saw 2 variants (Alpha and Delta), both extremely different from the 2020 classic version
- As if that were not enough, in the first 6 months of 2022 we’ve seen 5 distinct Omicron subvariants, each more infectious than the previous!
Conclusion: The virus is mutating extremely rapidly, adapting to its new human hosts (and, for that matter, wildlife outside Chinese bats). We must keep up with these variants, or accept a very high casualty rate in the near future.
Now, given the need not to mess up immunity to prior strains, Moderna went with a bivalent vaccine. Pfizer, for whatever reason, went monovalent: BNT162b2-OMI is against only the Omicron/BA.1 strain, then dominant. They tested it a couple of different ways:
- About 1200 patients got 3 shots of classic Pfizer, then divided into 2 arms getting either a 4th shot of classic Pfizer or the Omicron monovalent.
- A separate set of 205 patients got 2 primary doses of Pfizer Omicron monovalent as their first vaccine. (How did they find 200ish people who had refused vaccination up to now, but then agreed to be in a clinical trial? I have no idea.)
- They also experimented a bit with dose levels, looking at 30μg and 60μg. My guess is they got burned by too low a dose on their pediatric trials (and had to lobby for a 3-dose pediatric protocol), so now they’re unwinding a bit and exploring higher doses.
- Their analyses also stratified a bit by age, to show it didn’t go away for elders.
- Finally, they tested it against Omicron/BA.4-5, to see if it was still up-to-date.
The goals were the same as before: antibody levels showing GMR superiority (95% lower confidence limit of GMR > 1), and seroresponse noninferiority (95% lower confidence limit of SRR > -5%, i.e., guarantee little if any seroresponse loss) on Omicron, and at least comparable response on the reference strain.
Now, that’s a lot of different studies, which leads to a lot of different ways to compare the data! Would you believe me if I said it looked like all the side analyses were favorable, and just hit the summary? (If you answer “no”, then I remind you the full presentation is in the notes & references!)
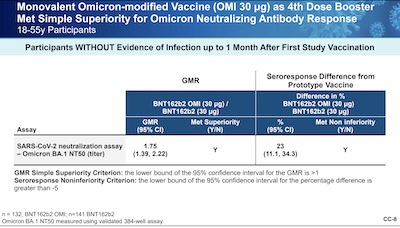
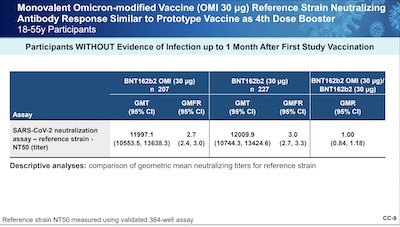
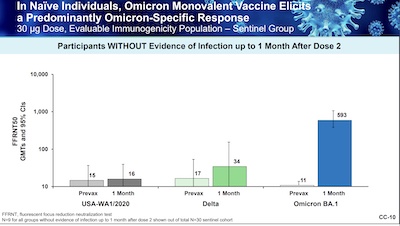 Slides 8 - 10 do the trick for getting the bottom line into our brains:
Slides 8 - 10 do the trick for getting the bottom line into our brains:
- Slide 8: They were supposed to show, against Omicron, superiority in antibody levels as measured by GMR and at least non-inferiority in seroresponse rates. In fact, they showed superiority in both: GMR ~ 1.75 and about 23% gain in seroresponse rate. Well done.
- Slide 9: With respect to the reference strain, they were supposed to show non-inferiority, and that has clearly been done, with a GMR ~ 1.00. (And they did finally add the 95% confidence limits that we missed above in the FDA report. Good.)
- Slide 10: All the above was for people who had previously gotten the classic Pfizer vaccine. What about immunologically naïve people who got only the Omicron version? This slide shows the somewhat annoying result: they got strong immunity to Omicron, but not to the previous variants! So basically in order to get the Pfizer monovalent vaccine to work well, you must have previously received the classic vaccine. This is why Moderna went with a bivalent formulation!
So this is pretty good: nice response in GMR and seroresponse percentages, provided you’d previously gotten the classic vaccine. So with Pfizer, there have to be 2 vaccines. Moderna’s bivalent can replace the previous Moderna vaccine, which Pfizer cannot do.
(And still no explanation of the weirdness in higher GMT levels reported by Pfizer.)
Novavax
Novavax [15] is a bit of an odd duck here. Although the VRBPAC recommended approval of their adjuvanted protein vaccine on 2022-Jun-07, to my knowledge the FDA has not yet accepted that recommendation, nor has the CDC ruled on it. Apparently there are changes to manufacturing that are causing hair to be pulled out. [16]
They made some interesting claims:
- Their recombinant trimeric spike protein displays conserved epitopes across variants.
- Their adjuvant causes “epitope spreading” and thus enhanced recognition of those conserved epitopes.
Therefore, they claim, their protein vaccine should be fine with Omicron.
I’m not quite sure what to make of this, and apparently neither were the VRBPAC members. In any case, their vaccine is not yet approved since they’ve changed their manufacturing pipeline to make something slightly different from what was in their clinical trials, so that has to be ironed out first with a bridging study.
Weird, but useful if true.
The WHO and FDA on variant booster composition
Lastly, there were presentations from the WHO on vaccine composition [17] and another presentation from the FDA on the same subject [18], apparently teeing up the discussion questions on vaccine strains.
This is all a bit frustrating to me. Moderna’s previously indicated there’s really not time to fuss about with the composition now in mid-summer, and still expect vaccines available in early fall. So pardon my head-banging while we look through these talks.
The WHO presentation was a bit long winded, essentially saying yet again that Omicron is the main strain that matters now, but broad protection is still needed, and vaccine efficacy is fading both with time and the evolution of Omicron. Ok, but we all know that!
In the end, they came down in favor of bivalent boosters as a way of punching Omicron in the nose while preserving some punches for the older strains. That’s… actually pretty sensible to me.
Weir’s FDA presentation, frankly, struck me as dithering about all the issues that might need to be considered. I suppose it does tee up the discussion nicely, so good for him. But I just find it tough to take that somebody might want to change the strains at the last minute and endanger the manufacturing schedule.
Discussion and voting
A few VRBPAC members were big fans of Novavax, calling it “the most compelling thing [they’ve] seen today”. I’m still kind of weirded out by it. But then again maybe I’m not enough of an expert here to be able to tell “weird” from “wonderful”.
There were also some complaints that there were no pediatric data on the new vaccines, though typically that would come later after the main vaccine is let loose.
There was also some concern about equity considerations world-wide, where the developing world will see the US changing the vaccine for itself but leaving none for anybody else. Yes, that’s a real concern, but it’s a concern about political, economic, and distribution policies, not about the science of whether this works at all.
The final vote on whether to recommend updated strains in the next round of boosters was 19 Yes, 2 No, and 0 Abstain. The no votes were from Paul Offit of Children’s Hospital of Philadelphia & Hank Bernstein of the Zucker School of Medicine.
- Offit had previously opined that we’re moving too fast and that variant vaccines are really new products that should go through the full-court press of a formal review like the original vaccines. He does, however, acknowledge that the existing vaccines given as boosters are still a pretty good fall-back position.
- I must have missed it if Bernstein said anything in particular. (At this point, my attention span was flagging.)
The Weekend Conclusion
- Moderna’s bivalent vaccine works, and works well. If I have a choice, this is the one I want to get in the fall. It could probably be used as a complete drop-in replacement for the original Moderna vaccine, though I haven’t seen that discussed.
- Pfizer monovalent vaccine also works, provided you’ve previously had the classic vaccine. Thus it’s not a replacement for the original Pfizer vaccine, but a booster to be used afterwards, probably at the 4th dose. (And there was no clarification why, in the table above, the GMT levels for Pfizer were an order of magnitude larger than Moderna’s. That gives me a statistical itch.)
- Novavax is weird, but some people are enthused. I’ll wait for that to shake out a bit more before having an opinion.
So the VRBPAC recommended the FDA proceed with Omicron-specific boosters. We’ll see what they do about it.
My opinion is that they should EUA them immediately, but then nobody has to care about the opinion of a cranky old scientist who’s the proprietor of a Crummy Little Blog That Nobody Reads (CLBTNR). :-)
Addendum 2022-Jun-29
Katelyn Jetelina at Your Local Epidemiologist has also written a summary of the VRBPAC meeting, coming to broadly similar conclusions. And much greater brevity!
Addendum 2022-Jul-02
Reuters reports [19] [20] [21] that the FDA (a) will require Omicron/BA.4 and BA.5 content in fall boosters, but (b) will not require new clinical trials, regarding the existing Omicron trials above as sufficient.
Now we’ll find out who’s right about whether the manufacturing pipeline can turn on a dime and produce vaccines in time for fall!
Notes & References
1: US FDA Staff, “FOOD AND DRUG ADMINISTRATION (FDA), Center for Biologics Evaluation and Research (CBER): 175th Meeting of the Vaccines and Related Biological Products Advisory Committee June 28, 2022 AGENDA”, US Food & Drug Administration, downloaded 2022-Jun-27. ↩
2: US FDA Staff, “Food and Drug Administration, Center for Biologics Evaluation and Research, Office of Vaccines Research and Review: 175th Meeting of the Vaccines and Related Biological Products Advisory Committee June 28, 2022, Meeting Roster”, US Food & Drug Administration, downloaded 2022-Jun-27. 16 members + 13 temporary voting members + 8 guest speakers + 4 FDA participants + 6 FDA administrative staff. ↩
3: US FDA Staff, “175th Vaccines and Related Biological Products Advisory Committee (VRBPAC) Meeting June 28, 2022: DISCUSSION QUESTIONS”, US Food & Drug Administration, downloaded 2022-Jun-27. ↩
4: US FDA Staff, “175th Vaccines and Related Biological Products Advisory Committee (VRBPAC) Meeting June 28, 2022: VOTING QUESTION”, US Food & Drug Administration, downloaded 2022-Jun-27. ↩
5: US FDA Staff, “FDA Briefing Document, Vaccines and Related Biological Products Advisory Committee Meeting June 28, 2022: SARS-CoV-2 strain composition of COVID-19 vaccines”, US Food & Drug Administration, downloaded 2022-Jun-27. ↩
6: Elise Meyer (Moderna Corporate Communications), “MODERNA ANNOUNCES OMICRON-CONTAINING BIVALENT BOOSTER CANDIDATE MRNA-1273.214 DEMONSTRATES SUPERIOR ANTIBODY RESPONSE AGAINST OMICRON”, Moderna Press Releases, 2022-Jun-08. ↩
7: Pfizer Media Relations, “Pfizer and BioNTech Announce Omicron-Adapted COVID-19 Vaccine Candidates Demonstrate High Immune Response Against Omicron”, Pfizer press releases, 2022-Jun-25. ↩
8: P Marks, “Considerations for Whether and How the COVID-19 Strain Composition Should be Modified”, US FDA VRBPAC, 2022-Jun-28. ↩
9: US FDA Staff, “Emergency Use Authorization for Vaccines to Prevent COVID-19: Guidance for Industry “, US FDA, 2022-Mar-31, revised 2022-May-25. ↩
10: CDR H Scobie, “Update on Current Epidemiology of COVID-19 Pandemic and SARS-CoV-2 Variants”, US CDC, 2022-Jun-28. ↩
11: LCDR R Link-Gelles, “Updates on COVID-19 Vaccine Effectiveness during Omicron”, US CDC, 2022-Jun-28. ↩
12: J Lessler, “Round 13: Planning scenarios projecting COVID-19 burden March 2022-March 2023 under current vaccination policy”, COVID-19 Scenario Modeling Hub, 2022-Jun-28. ↩
13: S Hoge, “mRNA-1273.214: Moderna COVID-19 Investigational Bivalent Vaccine (Original + Omicron)”, Moderna, 2022-Jun-28. ↩
14: K Swanson, “Pfizer/BioNTech COVID-19 Omicron-Modified Vaccine Options”, Pfizer, 2022-Jun-28. And did you know that Canterbury Tales was set as a series of stories told by travelleres fleeing a plague? ↩
15: GM Glenn, “Novavax, Inc.: Vaccines and Related Biological Products Advisory Committee June 28, 2022”, Novavax, 2022-Jun-28. ↩
16: Healthline Staff, “FDA Approval of the Novavax COVID-19 Vaccine Delayed by Manufacturing Changes”, Healthline, not obviously dated but sometime shortly after 2022-Jun-09. ↩
17: K Subbarao, “Technical Advisory Group on COVID-19 Vaccine Composition (TAG-CO-VAC) Interim statement on current COVID-19 vaccine composition”, World Health Organization, 2022-Jun-28. ↩
18: JP Weir, “COVID-19 Vaccine Strain Composition”, US FDA, 2022-Jun-28. ↩
19: M Erman & M Mishra, “US FDA wants COVID boosters targeting Omicron BA.4, BA.5 subvariants”, Reuters, 2022-Jun-30. ↩
20: M Erman, “FDA will not require clinical trial data to authorize redesigned COVID boosters -official”, Reuters, 2022-Jun-30. ↩
21: M Erman, “U.S. FDA to use existing Omicron booster data to review shots targeting new subvariants -official”, Reuters, 2022-Jun-30. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.