Readout of Moderna Bivalent Classic/Omicron Vaccine
Tagged:COVID
/
PharmaAndBiotech
/
Statistics
Today the Moderna trial of a bivalent classic/Omicron COVID-19 vaccine read out. Looks pretty good, so a regulatory filing with the FDA is pending.
Finally: An Omicron-specific vaccine
We’ve kvetched endlessly on this crummy little blog that nobody reads (CLBTNR) about the lack of a vaccine for current viral variants (now just the Omicron subvariants and nothing else). It seems silly to keep using a vaccine against a now-presumably-extinct variant from 2019-2020.

 However, today is a Very Good Day: we have some positive results on that front. The
primary source is of course the Moderna press release. [1]
Because we hate press releases, we’ll supplement with coverage from the always-excellent
Matt Herper at STAT News. [2]
However, today is a Very Good Day: we have some positive results on that front. The
primary source is of course the Moderna press release. [1]
Because we hate press releases, we’ll supplement with coverage from the always-excellent
Matt Herper at STAT News. [2]
The now-classic “Spikevax”, whose name we love, was initially know by the id mRNA1273. (I still have to suppress my reflex of immediately memorizing compound ids so I’ll be able to understand my colleagues. Having retired from pharma research, I don’t have to do that any more!) The new vaccine is bivalent: it contains both the classic Spikevax mRNA against the classic Wuhan virus, and an Omicron-specific variant. It is known as mRNA1273.214.
Previous bivalent COVID-19 vaccines from Moderna are now zombies, as they worked against things like classic + Beta, but Beta is now irrelevant. The virus was literally evolving faster than we could get vaccines through clinical trials!
The clinical endpoint of the trial was that it had to be better than mRNA1273: the geometric mean titer (GMT) of antibodies had to be greater with mRNA1273.214, and even more stringently, the lower confidence limit on the geometric mean titer ratio (GMR) had to be greater than 1. In other words, “do better than the original vaccine” and be really sure about that.
Result: Antibody levels were 8-fold above baseline (when used as a booster on previously vaccinated individuals).
- Against Omicron: GMR = 1.75 (97.5% CL: 1.49 - 2.04).
- Against classic D614G: GMR = 1.22 (97.5% CL: 1.08 - 1.37).
- One month later: Neutralizing GMT against both was still stronger for mRNA1273.214 than for mRNA1273.
- All other variants: Binding antibody titers were higher, with 95% confidence, against Alpha, Beta, Gamma, and Delta as well.
Note the use of an even tighter confidence limit than is traditional: 97.5% vs 95% may not seem like much, but it means they’re really, really, truly sure about this result.

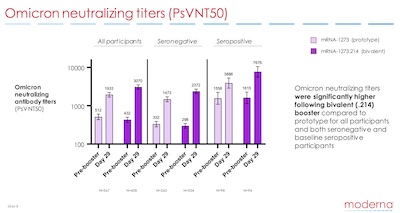 There was a corporate webcast, but I couldn’t bring myself to watch all the
corporate-speak. I have, however, archived their slide presentation here on this
CLBTNR. [3] Looking quickly through it (only 13 slides
including the usual corporate boilerplate nonsense, so really 10 meaningful slides), we
see it confirms the numbers in the press release with some more detail. Also, as shown
here on slide 8, it shows us the data behind the GMR result in terms of Omicron
neutralizing titers.
There was a corporate webcast, but I couldn’t bring myself to watch all the
corporate-speak. I have, however, archived their slide presentation here on this
CLBTNR. [3] Looking quickly through it (only 13 slides
including the usual corporate boilerplate nonsense, so really 10 meaningful slides), we
see it confirms the numbers in the press release with some more detail. Also, as shown
here on slide 8, it shows us the data behind the GMR result in terms of Omicron
neutralizing titers.
Moderna plans a filing with the FDA “in the coming weeks”. On the other hand, the FDA meets on 2022-Jun-28 to discuss which variants ought to go into a new vaccine. This is bitterly ironic: Moderna (and presumably Pfizer/BioNTech) have said there’s not enough time to switch now and expect vaccines available in the fall. So I hope we manage not to trip over our own tongues, and go along with mRNA1273.214!
The Weekend Conclusion
So, yeah: pending seeing the actual technical docs, this sure looks like it’s superior to the classic vaccine.
I want this thing. I want it for myself, for my family, for my friends, and for all of humanity. We should all live, and not die.
Notes & References
1: E Meyer, “MODERNA ANNOUNCES OMICRON-CONTAINING BIVALENT BOOSTER CANDIDATE MRNA-1273.214 DEMONSTRATES SUPERIOR ANTIBODY RESPONSE AGAINST OMICRON”, Moderna Press Releases, 2022-Jun-08. ↩
2: M Herper, “Moderna says Omicron-containing booster outperforms current vaccine”, STAT News, 2022-Jun-08. ↩
3: Moderna Staff, “Bivalent COVID Booster Ph 2/3 Interim Analysis (mRNA-1273.214)”, Moderna press materials, 2022-Jun-08. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.