FDA VRBPAC Meeting: Novavax COVID-19 Vaccine
Tagged:COVID
/
PharmaAndBiotech
/
Politics
/
Statistics
Today the FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) met to advise the FDA on whether to approve Novavax’s more traditional protein (non-mRNA) vaccine against COVID-19. Want to know who said what?
Why another vaccine, so late in the day?
First, enough with the “late” stuff. It’s only 2 1/2 years since the outbreak was widely recognized, around January 2020. In the past, vaccines have taken about 10 years to develop. Now we’re all spoiled with the absolutely amazing speed of the Pfizer/BioNTech and Moderna mRNA vaccines. But this is a more traditional protein-based vaccine, growing the spike protein in a culture of insect cells (used to be chicken eggs, now it’s usually some kind of caterpillar cell culture). And it only too 2.5 years, not 10. So cut them a break.
Ok, not “late” per se, but why another? As near as I can make out, 3 reasons:
- General diversification principle: we want as many different ways of fighting the pandemic as we can get, in the event we have to drop some for safety or just supply chain failure.
- mRNA Vaccine holdouts: As a traditional protein vaccine, this may lure in some vaccine hesitant persons who attach too high an “ick” factor to mRNA vaccines. I’m suspicious of this, as it seems most vaccine resisters are pretty dug in at this point and are likely unreachable. Still, there’s a chance, so why not take a chance that has little downside and might save the lives of a few stubborn holdouts?
-
Cold chain requirements: mRNA is a ridiculously fragile molecule, initially requiring storage at -80°C – -60°C which is rare outside big city hospitals and medical research institutions, and unattainable in the developing world. (It’s slightly better now.)
But NVX-CoV2373/Nuvaxovid can be stored at +2°C – +8°C (albeit in purpose-built vaccine refrigerators). This is much more manageable, especially in the developing world.
 I’m not the only one wondering about this; just last week there was an article in the
New England Journal of Medicine also asking whether another vaccine is
needed! [1]
I’m not the only one wondering about this; just last week there was an article in the
New England Journal of Medicine also asking whether another vaccine is
needed! [1]
Some of their arguments:
- It looks like we’re on track to get 70% vaccination world-wide by the end of 2022. (Though it seems to me that herd immunity to Omicron is going to need > 90% vaccination?)
- There are at least 344 COVID-19 vaccines developed or in late development.
- There are 31 vaccines already in large-scale use in some country.
- They use a variety of platforms: mRNA, viral vector, inactivated whole-virus, protein subunits, and plasmid DNA are the ones that come to mind. Never in human history have we done anything like that!
- But, considerations beyond efficacy and safety apply: ease of schedules, integration with routine vaccinations, need for boosters, cost, cold-chain logistics, scaling of manufacturing, community acceptance, and possibility of local production.
All excellent points, and illustrate several reasons I hadn’t thought about. Yes, the newer vaccines tend to have efficacies ranging from 69% – 85%, and that’s less than the mRNA vaccines at 95% or so. But if you can’t get the mRNA vaccine in position because of cold chain problems, or people won’t accept it because of mRNA distrust, then the next-best vaccine is the way to save lives.
Remember, with a pandemic, nobody is safe until everybody is safe. We need to care about the developing world because it’s what decent people do, but it’s also in our interest to suppress the proliferation of new variants.
A quick early look
The FDA VRBPAC meeting page [2] has links to all the meeting materials. The voting question [3] is short and sensible:
Based on the totality of scientific evidence available, do the benefits of the Novavax COVID-19 Vaccine when administered as a 2-dose series outweigh its risks for use in individuals 18 years of age and older?
Last Friday, the Novavax briefing document and the FDA briefing document became suddenly available (though not much else). [4] [5]
The slide presentations will have all the good stuff, but the briefing docs have at least
a hint at what’s going down:
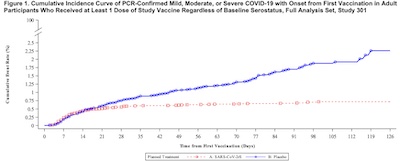
- First comes the now-expected beautiful Kaplan-Meier curve (p. 26 in the FDA document), shown here. The blue curve is the unvaccinated population, and the red is the vaccinated. The effect doesn’t kick in until about day 20 or 28 after the first dose, but the red curve does flatten admirably.
- Next, vaccine efficacy (p. 23ff in the FDA document):
- All participants: 90.41% (CL: 83.81% – 94.32%)
- Age 18 - < 65 yr: 91.06% (CL: 84.44% – 94.87%)
- Age ≥ 65yr: 78.63% (CL: -16.64% – 96.08%)
The prespecified success criterion is the same as was given 2 years ago for the mRNA vaccine efficacies: median(VE) ≥ 50% and lower confidence limit LCL(VE) ≥ 30%. So everything looks pretty good, except the age 65+ cohort, where we don’t meet the LCL criterion! That’s a problem, but:
- There were only 6 cases in this cohort (2/2044 vax vs 4/968 placebo), which makes the confidence limits very, very wide.
- The FDA requested post-hoc analysis of antibody levels in the 50-64 vax arm and the 65+ vax arm. The ratio was 0.91 (CL: 0.68 – 1.2), so the seniors do have pretty significant antibodies, roughly comparable with their juniors.
So we might try to assign the alarming lower confidence limit in elders to rare events and take comfort in the comparable geometric mean titer antibody levels (both good things).
The other issue is there were 4-5 cases of myocarditis or pericarditis in the vax arm. (Novavax says 4, the FDA says 5 including one case whose symptoms were likely myocarditis.) The mRNA vaccines have seen this post-approval, but the rate was low enough that it wasn’t really seen much in the trials. So that’s potentially a stumbler.
In adverse events, the Novavax document reports “gin shot wound” (p. 59, Table 15), by which I think they mean “gun shot wound”! These documents are gone over so carefully by so many people, I’m slightly surprised at the spelling gaffe. Still, gunshot is unlikely vaccine related. (Previously, we blogged about how Moderna had to report getting struck by lightning for one participant. The FDA sensibly gave them a pass on that one.)
So it might get approval pretty quickly, or there might be a food fight because we already have such good mRNA vaccines.
Honestly, I couldn’t predict which.
That’s why they have the meeting.
Agenda and conflicts of interest
The agenda [6] looks pretty straightforward:
- the usual welcome and introductions,
- a formal statement of the EUA question by the FDA,
- a couple CDC presentations reminding us we still have to take COVID-19 seriously and myocarditis is potentially a risk,
- the Novavax presentation analyzing their trial results,
- the FDA presentation independently analyzing the same data,
- some public hearings which are always skippable,
- discussion & voting.
Slightly more interesting is the waivers for potential conflicts of interest, almost always because they need some expertise from industry or some doctor happens to have some relevant investments [7]:
No waivers were issued for conflicts of interest for this meeting
 I’ve never before seen them say there were no conflicts of interest. Or, more
technically, no waivers for such conflicts were issued, so if there is one then I guess it’s on
the person with the conflict, not the FDA?
I’ve never before seen them say there were no conflicts of interest. Or, more
technically, no waivers for such conflicts were issued, so if there is one then I guess it’s on
the person with the conflict, not the FDA?
Hmpf. Maybe I’m too suspicious.
Our usual safari guides (via an unusual channel)
No liveblog at STATNews today, but Helen Branswell is live-tweeting, if you want to follow along for another perspective:
Setting the stage
 First up is a presentation by Goutam Sen [8], documenting:
First up is a presentation by Goutam Sen [8], documenting:
- the general situation of the pandemic,
- what the Novavax vaccine is,
- what the considerations for an EUA are,
- what other vaccines are available in the US,
- the meeting agenda, and
- the all-important voting question above.
The content was unsurprising, generally. But a few things struck me oddly:
- Novavax contains the spike protein for the original Wuhan strain, not anything more recent. That’s what I expected, but I’m still impatient for an Omicron-specific vaccine.
- It’s cultured in Sf9 cells, a cell culture derived from the caterpillars of a certain moth. They take transfection of a foreign gene – say, the spike protein – from a baculovirus which people like to use in the lab because it cannot replicate in mammalian cells. The point of interest: people are squeamish about mRNA from a synthesizer, but not about proteins generated in a viral-transfected insect cell culture?!
- It also uses a saponin-based adjuvant (Chilean soap tree extract and some bacterial surface antigens), which is basically something to irritate the immune system into reacting more strongly to the spike protein. This is standard.
- The EUA application was received 2022-Feb-01. Why are we only acting on it now?!
CDC on the public health situation
 CDR Heather Scobie (it still freaks me out that the US Public Health Service) uses
military ranks!) presented next on the general state of COVID-19 epidemiology and
vaccination in the US. [9]
CDR Heather Scobie (it still freaks me out that the US Public Health Service) uses
military ranks!) presented next on the general state of COVID-19 epidemiology and
vaccination in the US. [9]
This turned out to be really interesting, giving a picture of where we are with
vaccination and viral variants that is seldom put together in one place like this. Summary:
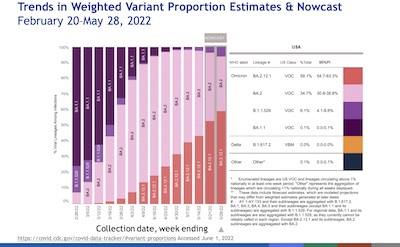
- All Omicron, all the time. It used to be all Delta, all the time. Now Omicron has
bigfoot-stomped Delta into extinction, and Omicron sub-variants are competing to
bigfoot-stomp each other out of existence. I never thought this would get to be as
contagious as measles, but… nonetheless, here we are. Her slide shown here tells
us graphically that nothing other than Omicron variants matters; BA.2.12.1 is likely to
dominate soon. (After that, BA.4 and BA.5 out of South Africa?)
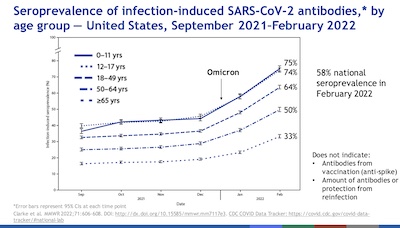
- As we’ve mentioned before, you can
measure the population who have been infected (even briefly and asymptomatically) with
the virus by looking at oddball antibodies, like the seroprevalence of the nucleocapsid
antibody. The CDC is, unsurprisingly, on top of this. The evidence is that 58% of the
US population has had some level of COVID-19 infection in the past, though for most of
them vaccination has helped them clear it quickly. As you can see here, it’s highly
stratified by age, with 0-18 years being by far the dominant subgroup.
- Basically, kids go everywhere, congregate in groups, and you can’t impose 100% effective masking or social distancing on them.
- Also, among the elderly, the death rate is higher: they’re not around for
retrospective blood draws to see if they were previously infected.
Talk about inter-generational conflict!
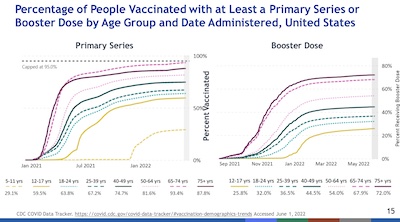
- Vaccination rates in the US have definitely reached their plateau, as seen here by the
plots of vax and boost rates vs time, stratified by age. Again, age group seems to be
the driving factor. I wish I knew what we could do to raise the plateau of younger
people!
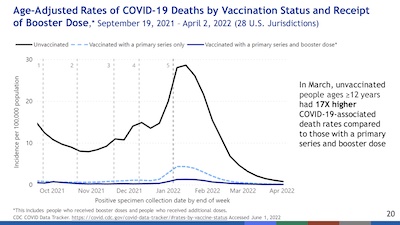
- Next, consider death rates. Here Scobie shows us death rates by vaccination status,
normalized by the age-adjusted population size for each vax status. I often say
metaphorically that something is “brutally clear”, but this is not metaphorical. The
death rates are almost exclusively the unvaccinated, tiny among the vaccinated, and
almost negligible among the boosted. Refusing vaccination/boosting is volunteering to
die, and attempting to take down the rest of society with you.
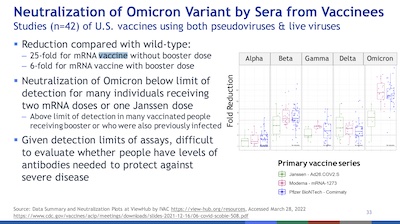
- Finally, let’s consider vaccine efficacy as the virus has evolved. What the plot shows
here is a fold reduction in how well vaccine sera neutralize the variants. That is,
higher is worse: 2x means the vaccine sera are 1/2 as effective. The fold reduction in
efficacy generally rises with later variants. The curious part is the J&J vaccine,
which did better? No idea what’s going on there!
- Note that the graph shows only for vaccination without boosting.
- But boosting works: 25x weaker without boosting becomes only 6x weaker with boosting.
- However, 6x weaker is no fun: we need, apparently, variant-specific vaccines now. I wish I understood the hold-up in the Omicron-specific vaccines, but that will apparently be debated at a VRBPAC toward the end of this month.
 Next CAPT Tom Shimabukuro (again, military rank in US Public Health Service!) presented on
the state of myocarditis in relation to mRNA COVID-19 vaccines. [10]
The relevance, of course, is to compare with myocarditis in the Novavax vaccine, about
which more later.
Next CAPT Tom Shimabukuro (again, military rank in US Public Health Service!) presented on
the state of myocarditis in relation to mRNA COVID-19 vaccines. [10]
The relevance, of course, is to compare with myocarditis in the Novavax vaccine, about
which more later.
 I got rather less out of this, other than:
I got rather less out of this, other than:
- While there is now evidence of a causal relation between mRNA vaccination and myocarditis, it happens at a pretty low rate and that rate is lower than the rate caused by actually getting COVID-19 (see table). So you’re still better off vaccinated.
- It’s primarily, though not completely, a phenomenon in younger males, who tend to recover pretty nicely. Better recovery, in fact, than if they’d had COVID-19.
The Novavax presentation & the FDA presentation

 This presentation [11] has all the goods, but it’s a bit
verbose in the way that multi-presenter slide decks sometimes get. I’m just going to hit
the high points from both this and the FDA presentation on the same
data. [12]
This presentation [11] has all the goods, but it’s a bit
verbose in the way that multi-presenter slide decks sometimes get. I’m just going to hit
the high points from both this and the FDA presentation on the same
data. [12]
There’s (appropriately) a lot of stuff about the design of the clinical trials: Phase I in Australia ($N = 29$), Phase 2 in South Africa ($N = 2211$, including HIV+), Phase 2 in the US & Australia ($N = 513$), Phase 3 in the UK ($N = 7569$), and another Phase 3 in the US ($N = 19735$). So lots of people: at least 27k in the combined Phase 3’s alone. Sure, Pfizer & Moderna had like 40k, but this is quite sufficient to prove the point.
Alas, however: the clinical data is from almost entirely before Delta, let alone before Omicron. So the application to the current situation may be questioned; this is the price of moving slowly against a virus that evolves quickly.
Also, there seem to have been enough manufacturing problems that the vaccine going forward may not be entirely identical with the vaccine used in the trial. Novavax says they have enough compatibility data between old & new versions; the FDA thinks otherwise. This would normally be a huge black flag dooming the project, but apparently not here.
Also also, everybody wonders what “emergency use authorization” means, when there are 2 vaccines that are fully approved and already in place. VRBPAC member Eric Rubin in fact brought this up at the start of the meeting; apparently the statutes allow a lot of leeway for “unmet medical need.”
So, plenty to wonder about.
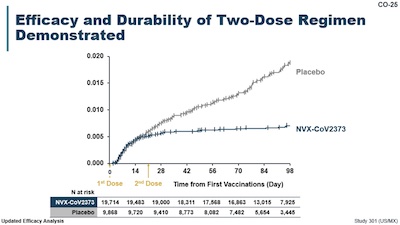 Why bother with the heavy lift of all that wondering? Pretty much the beautiful KM curve
from Novavax slide 25, shown here:
Why bother with the heavy lift of all that wondering? Pretty much the beautiful KM curve
from Novavax slide 25, shown here:
- The horizontal axis is time, and the vertical axis is the fraction of patients getting COVID-19.
- The blue curve is the vaccinated patients, and the gray curve is the placebo patients.
- The vertical marks on each curve are censorship events, when a person drops out of the trial. Cox proportional hazard regression was invented pretty much to handle that properly. (We can’t reproduce it with the data in the paper, so we’ll use cruder methods.)
The brutally obvious fact, of course, is that the vaccinated participants clearly did better. There are ways to quantify that, e.g., with hazard ratios and logrank $p$-value tests, and they do that, but it’s clear visually here.
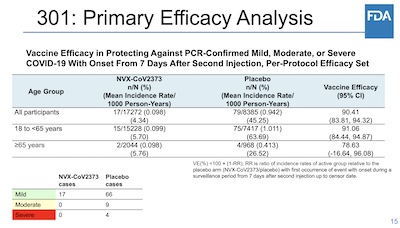 The other way to quantify that the vaccinated participants did better is by calculating
vaccine efficacy and its 95% confidence limits. We’ve gone over that ground before, so we
won’t do that again here. But slide 15 of the FDA presentation makes a powerful case that
the efficacy is generally high and well bounded above the 30% threshold demanded by the
FDA.
The other way to quantify that the vaccinated participants did better is by calculating
vaccine efficacy and its 95% confidence limits. We’ve gone over that ground before, so we
won’t do that again here. But slide 15 of the FDA presentation makes a powerful case that
the efficacy is generally high and well bounded above the 30% threshold demanded by the
FDA.
Can we check the FDA/Novavax efficacy numbers and their confidence limits with our cruder scaled binomial confidence interval method? [13] Why yes, yes we can:
> source("../assets/2021-11-12-covid-treatments-simple-efficacy-confidence-limits.r")
Loading required package: ggplot2
Learn more about the underlying theory at https://ggplot2-book.org/
> signif(efficacyAndCL(17272, 17, 8385, 79), digits = 3) # all participants
LCL Eff UCL
0.825 0.896 0.938
> signif(efficacyAndCL(15228, 15, 7417, 75), digits = 3) # 18 to < 65 years
LCL Eff UCL
0.832 0.903 0.944
> signif(efficacyAndCL(2044, 2, 968, 4), digits = 3) # >= 65 years
LCL Eff UCL
-0.104 0.763 0.949
Now, our numbers don’t match the FDA’s exactly, but then the FDA and Novavax did a sophisticated Cox regression taking into account censorship while we did not. Generally, we’re within about 1% of each other, with my crude estimates being generally lower. (The exception is the miserable lower confidence limit for seniors, where they got -16% and I got -10% with cruder methods. Broadly, we agree: there were probably too few cases in this age cohort to measure very well and thus we got a super-wide confidence interval. But the rest of the cases look eminently plausible.)
 Now, you can’t just leave things like that: people will want to know if the vaccine really
failed for age 65+, or if there were just too few deaths in both arms to get good
statistics. So the FDA asked Novavax to go measure antibodies in the blood of people in
the treatment arm, grouped by age. When you take the ratio of the geometric mean titer
antibody levels, do you find the seniors are way lower, or comparable?
Now, you can’t just leave things like that: people will want to know if the vaccine really
failed for age 65+, or if there were just too few deaths in both arms to get good
statistics. So the FDA asked Novavax to go measure antibodies in the blood of people in
the treatment arm, grouped by age. When you take the ratio of the geometric mean titer
antibody levels, do you find the seniors are way lower, or comparable?
The results are shown here on slide 16: the seniors have 91% of the antibody levels of their juniors (CL: 68% - 120%). This gives one confidence that immunity really was stimulated in the elder cohort, and the wide confidence interval for efficacy was just because too few people died in both arms.
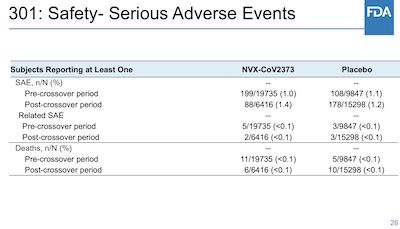 Next, there’s a lot of analysis about adverse events. I don’t really have the patience to
wander through it all, but liked this summary on slide 26 from the FDA: serious adverse
event frequencies in both arms are pretty comparable. (Though the gunshot wound and the 2
deaths from motor vehicle accidents were in the treatment arm, though nobody’s fault.
It’s weird stuff like this that tells you the data is real. Nobody fakes weird junk like
this when they fake data!)
Next, there’s a lot of analysis about adverse events. I don’t really have the patience to
wander through it all, but liked this summary on slide 26 from the FDA: serious adverse
event frequencies in both arms are pretty comparable. (Though the gunshot wound and the 2
deaths from motor vehicle accidents were in the treatment arm, though nobody’s fault.
It’s weird stuff like this that tells you the data is real. Nobody fakes weird junk like
this when they fake data!)
That’s generally reassuring.
There was a lot of arguing back and forth about myocarditis, pericarditis, and so on. Some thought it was causally related to the vaccine, some not. What pushed me over the hump toward “approve and move on” was this statement on slide 37 from the FDA:
In a total clinical safety database of ~ 40,000 vaccine recipients, to date, 6 NVX recipients have reported myocarditis and/or pericarditis, including 5 events within 20-days post-NVX.
Basically, causally linked or not, it’s rare. Much rarer than the various dangers posed by COVID-19, so the risk trade-off is overwhelmingly in favor of vaccination.
Discussion & Voting
Confession time: I went to buy groceries. Again. So I didn’t listen to all the details here. Also, I deliberately skipped the public comment section, because that gives me nightmares. (Literally, if you need to know.)
There was apparently some arguing about myocarditis. NVX says no relation, the FDA said “maybe”. Dorian Fink of the FDA said “maybe” is enough to add myocarditis to the warnings, so they should do that if they approve it. Seems like a reasonable compromise, or as reasonable one as you’re likely to get.
Bruce Gellin (former head of HHS national vaccine program, now Chief of Global Public Health Strategy, The Rockefeller Foundation had some interesting not-quite-accusations:
- Novavax has more data than they’ve shown the FDA, so he wants to know what’s in there.
- He wants to know more about the different versions due to the manufacturing problems. He doesn’t buy that Novavax has convincing compatibility data between them, so we’d be approving a different vaccine than the one tested.
- He also wants to know the efficacy vs Omicron, since this was tested almost entirely before even Delta, let alone Omicron.
All reasonable questions, though it seems to me unreasonable to block the vaccine based on them. More apt would be to demand post-approval surveillance to patch up the compatibility data and get Omicron efficacy numbers, and demand disclosure of this other data about which I know nothing.
In the end, the vote was 21 yes, 0 no, and 1 abstention. The abstention was Gellin, for the reasons above. He said he wanted to vote a “conditional yes”, but that wasn’t an option.
So… stamp an EUA on it and get the CDC on the stick.
The Weekend Conclusion
Man, these folk love calling each other “doctor”! In my corner of the world, where we have mere PhDs, nobody does that. In fact, friends who have PhDs but work with clinicians are always careful to warn you should get your lab coat to say “Dr. Your Name”, and never “Your Name, PhD”. The latter will ensure that no MD will ever listen to anything you say. Medicine is very authoritarian compared to physics & math! I wonder if that will change with the next generation?
The efficacy data looked plenty good enough, and the safety data looked reasonable. So approval was reasonable.
I’m skeptical it will win over the vaccine defiant who get icked out by mRNA. What will they say when they find out it’s grown in insect cell cultures infected by a genetically modified virus? (That’s true of almost any other mass-produced virus too, but rationality doesn’t seem to be the sticking point here.)
The less demanding cold chain will be a good thing, especially in the developing world. Of course, the FDA has nothing to say about the developing world, but perhaps their imprimatur will help?
“Perhaps X will help” is weak sauce, bit seems to be what’s on offer in a world where people refuse the vaccines made available for a year and a half now.
This is a really weird timeline. Can I get back to the one I used to know?
Addendum 2022-Jul-20: CDC’s ACIP Approves Novavax
On 2022-Jul-20, epidemiologist Katelyn Jetelina reports at Your Local Epidemiologist that the CDC’s ACIP committee has finally approved Novavax on 2022-Jul-19! [14]
While that’s a good thing, I suppose, one wonders why they took 42 days to do so?! That’s pretty relaxed, even by their standards.
Everybody was hoping this would lure in a few more vaccine resisters, but… I think they’re pretty dug in with the anti-vax pit they’ve dug themselves. True, Jetelina points to a CDC survey where 16% of the unvaccinated said they’d get Novavax, but a more recent one said 10%. Now, figure there are 25% unvaccinated, so 10% of that is… 2.5% of the US population. Epidemiologically, that’s meaning-free.
The cold chain requirements are better, but in the developed nations we’ve solved that. Trying to get developing nations to take Novavax will definitely look like a class distinction that everybody will resent, whether that’s true or not.
Something just isn’t right here.
Notes & References
1: H Nohynke, et al., “Does the World Still Need New Covid-19 Vaccines?”, NEJM 386:2140-2142, 2022-Jun-02. DOI: 10.1056/NEJMe2204695. ↩
2: US FDA Staff, “Vaccines and Related Biological Products Advisory Committee June 7, 2022 Meeting Announcement”, US FDA, 2022-Jun-07. Contains meeting presentation materials and both FDA & Novavax analysis documents. ↩
3: US FDA Staff, “173rd Vaccines and Related Biological Products Advisory Committee (VRBPAC) Meeting, June 7, 2022: VOTING QUESTION”, US FDA, 2022-Jun-07. ↩
4: FDA Staff, “FDA Briefing Document: Novavax COVID-19 Vaccine”, US FDA, 2022-Jun-07. ↩
5: Novavax Staff, “NVX-CoV2373 Vaccine for the Prevention of COVID-19”, Novavax, 2022-Jun-07. ↩
6: FDA Staff, “173rd Meeting of the Vaccines and Related Biological Products Advisory Committee: June 7, 2022, DRAFT AGENDA”, US FDA, 2022-Jun-07. NB: It says “draft” agenda, but I’m looking at it while the meeting is happening, so as drafts go it’s pretty dang authoritative. ↩
7: FDA Staff, “Waivers for Conflicts of Interest”, US FDA, 2022-Jun-07. ↩
8: G Sen, “Novavax COVID-19 Vaccine, Adjuvanted: Request for Emergency Use Authorization”, US FDA/CBER/DVRPA, 2022-Jun-07. ↩
9: H Scobie, “COVID-19 Epidemiology and Vaccination Rates in the United States”, CDC COVID-19 Epidemiology Task Force, 2022-Jun-07. ↩
10: T Shimabukuro, “Update on myocarditis following mRNA COVID-19 vaccination”, CDC COVID-19 Vaccine Coordination Unit, 2022-Jun-07. ↩
11: F Dubovsky, R Mallory, D Kim, GA Poland, “Emergency Use Authorization (EUA) Application for NVX-CoV2373”, Novavax, 2022-Jun-07. ↩
12: L Lee, “FDA Review of Effectiveness and Safety of Novavax COVID-19 Vaccine in Adults ≥ 18 Years of Age Emergency Use Authorization Request”, US FDA Office of Vaccines Research and Review, 2022-Jun-07. ↩
13: Weekend Editor, “R script for efficacy and confidence limits using a scaled binomial model”, Some Weekend Reading blog, 2021-Nov-12. ↩
14: K Jetelina, “Novavax is here! Just not the silver bullet we need”, Your Local Epidemiologist blog, 2022-Jul-20. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.