FDA Considers COVID-19 Boosters for Pfizer Comirnaty
Tagged:COVID
/
Math
/
MathInTheNews
/
PharmaAndBiotech
/
SomebodyAskedMe
/
Statistics
Today the FDA’s Vaccines and Related Biological Products Advisory Committee meets to review the Pfizer/BioNTech application for 3rd dose boosters of their COVID-19 vaccine, Comirnaty. Opinion is divided, so there will be some arguing. I’m makin’ popcorn.
What’s the Sitch?
So… boosters, huh?
Last night somebody asked me about my position on boosters. Briefly: the evidence is somewhat ambiguous (as Simpson’s Paradox showed in the Israeli data).
- It seems clear that the immunocompromised (immune disease sufferers, those taking immunosuppressants for transplants, etc.) should get a booster, since they probably never got much of an immune reaction in the first place.
- Second, it seems similarly clear that the elderly should also get a booster, since their immune systems take a bit longer to respond, and respond more weakly. (The flu shot I just got had the “geezer dose”: 4x the dose for the youngs. Take that, kiddos!) Where to draw the age line for boosters is of course up for grabs; I’d be surprised if it were lower than 65 or higher than 75.
Basically, a booster won’t hurt, and will help any given individual. Whether that’s the best course or not for humanity as a whole is up for debate. We are probably better off using those doses to vaccinate the hesitant/resistant/defiant holdouts in the US, and everybody else in the world to stop the evolution of new variants.
That being said, if I’m offered a booster, I’ll take it. My refusal would (a) be modeling incorrect behavior for the vaccine hesitant/resistant/defiant, and (b) not guarantee my dose would go to an unvaccinated person, but instead just that somebody else would get a booster.
So that’s my prediction, if we’re being sensible: boost the immunocompromised, the elderly, and those at high risk (maybe like healthcare workers?); then concentrate the rest on the vaccine hesitant/resistant/defiant and the rest of the world. But as you know, the right-wing half of the US population cannot be described as anything even vaguely adjacent to “sensible”. (We used to tell people to “avoid something like the plague”, but it turns out Republicans don’t do that. Who knew?)
Today’s FDA VRBPAC Meeting on Boosters
Fortunately for all of us, my opinion is thoroughly irrelevant and about to become even more so. Today the FDA’s Vaccine and Related Biological Products Advisory Committee (VRBPAC) meets to consider the Pfizer/BioNTech application for authorization of 3rd shot boosters. Whether rationally concluded or not, that will settle the matter for a while.
 The FDA announced this meeting at the beginning of this month [1],
so things are moving very fast by their standards. (Anybody who tells you the FDA is
dragging their feet on COVID-19 vaccines doesn’t know how this works! Let alone why it
works that way.) Peter Marks, head of the FDA Center for Biologics Evaluation and
Research (CBER), said more or less the expected:
The FDA announced this meeting at the beginning of this month [1],
so things are moving very fast by their standards. (Anybody who tells you the FDA is
dragging their feet on COVID-19 vaccines doesn’t know how this works! Let alone why it
works that way.) Peter Marks, head of the FDA Center for Biologics Evaluation and
Research (CBER), said more or less the expected:
- The Biden administration announced a plan for acquisition & logistics of booster doses this fall, and
- FDA evaluation and approval is a prerequisite for that, and
- A data package submission is a prerequisite for that, and
- Pfizer/BioNTech have submitted such a package so they’ll be moving it forward.
Basically: “We’re all doin’ that thang we’re s’pposta do! Nothin’ t’see here kid. Move ‘long.”
It is of course being livestreamed direct from the FDA to various news media and the FDA’s own YouTube account (which I’ve embedded here).
 The meeting agenda [2] looks pretty straightforward, too.
The meeting agenda [2] looks pretty straightforward, too.
- Random welcome/roll call/introduction stuff that is de rigeur in these sorts of meetings,
- FDA backgrounder, CDC summary of the epidemiology of Delta and breakthrough infections, some real-world data on vaccine efficacy across the world,
- Sponsor presentation by Pfizer/BioNTech,
- FDA presentation giving their independent evaluation of the same data,
- Lunch (only 25min, so I hope somebody brings them sandwiches!), public hearings (usually pretty uninformed, ranty & not worth the time, but legally required),
- And then the important part: Q&A where the committee grills the submitter followed by discussion and voting.
It all looks pretty reasonable, though given the high level of disagreement and tempers involved, it could get a little heated. I hope not.
The Data Packages
We could listen to the newsies and their horse-race commentary, who’s up & who’s down, how important this or that person is, what the polls will say about politics and Biden’s policies… but I just don’t have the stomach for that. I mean, I’ll watch a bit of the expert testimony because those folks actually know what they’re talking about. But the newsies almost never know what they’re talking about, and insist on talking anyway. (Notable exception: STAT News, especially reporters like Helen Branswell & Matthew Herper. Below we will be well-guided by their liveblog of today’s events.)


 So instead we’ll look at the documents themselves that were submitted to the FDA.
Nothing like seeing the primary literature:
So instead we’ll look at the documents themselves that were submitted to the FDA.
Nothing like seeing the primary literature:
- Pfizer/BioNTech submitted a 53 page packet [3], which is surprisingly short as these things go. Interestingly, if you pick apart the PDF file the internal timestamps say it was last revised 2 days ago on 2021-Sep-15. Given that Pfizer/BioNTech submitted on 2021-Aug-25 [4], this surprised me. Must be a revised document, part of a “rolling submission”.
- As is customary in these matters, when you submit such a package to the FDA the first thing they do is throw away your conclusions and reanalyze the data themselves, using the methods specified in the proposal. Then they compare your answer to theirs, and look for any glaring inconsistencies. Only when they broadly match do you go ahead to the committee meeting like today (otherwise you get a sternly worded letter telling you to withdraw your application immediately or they’ll kill it forever and you won’t be able to resubmit!). So the FDA submitted their own 23pp package [5] which does exactly that.
- It looks like there are 7 other presentations on:
- real-world effectiveness of the vaccines across the world,
- the formal licensure application by the FDA,
- the FDA’s review of the safety and efficacy of boosters,
- Pfizer/BioNTech’s review of the same thing,
- a review of the booster data from the Israeli experience,
- a summary of the epidemiology of the Delta variant and its impact on the need for boosters,
- the supplemental Biologics License Application (BLA), which is somehow different from the formal licensure presentation.
So… lots of stuff! Being on one of these committees is work.
A Quick Look Through the Pfizer Package & Slides
 I didn’t scrutinze each of the 53 pages, but it seems they wanted to draw 2 major
conclusions:
I didn’t scrutinze each of the 53 pages, but it seems they wanted to draw 2 major
conclusions:
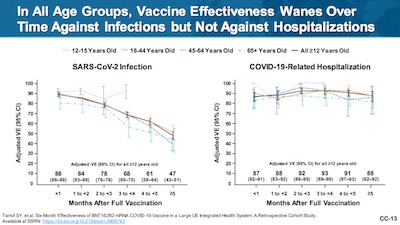
- A Kaiser Permanente of Southern California study showed vaccine efficacy waning (Figure 1, p. 12, makes this point, but slide CC-13 of their presentation [6] makes the point better and is reproduced here: the right plot shows sustained efficacy versus hospitalization). I’m glad they honestly say that efficacy against hospitalization and death remained high (mostly > 90%). So that’s sort of wanting to have things both ways: yes, efficacy against any infection might decline (though they don’t seem to be aware of Simpson’s paradox!), but the efficacy we care about is still robust. Hmpf.
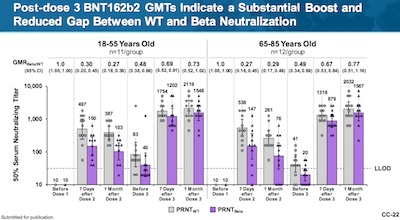
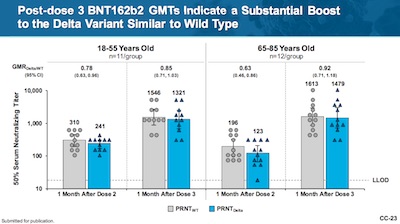
- A 3rd dose does indeed boost the geometric mean titer of antibodies, against both the wild-type and B.1.351 (Beta) variants (Figure 2, p. 19, makes this point, but slides CC-22 and CC-23 of their presentation make it more graphically, and include Delta). The rise after the 3rd dose is statistically significant, as shown by the confidence intervals. The 3rd dose is the rightmost 2 pairs of bars in each group. Note the vertical scale is a log scale, so these are really huge differences.
So… yeah, it works. But the evidence of need is slightly sketchy.
The FDA slide deck makes substantially the same points. [7] There’s some slightly subtle foofraw about noninferiority criteria according to the 97.5% lower confidence limit of the difference of… something or other about which you probably don’t care. While the details here are unlikely to be of broad interest, the game is to make sure the 3rd dose doesn’t harm immunity, and after that it helps.
One thing that stood out to me in the FDA slides is that this was a small study: $N = 23$ in Phase 1 and $N = 306$ in Phase 2/3. So it’s definitely underpowered to detect rare adverse events like the myocarditis/pericarditis in young males. Still, one can perhaps extrapolate from the rates seen in the first 2 shots, and hope the 3rd doesn’t change them for the worse.
Is Vaccine Efficacy Really Waning?
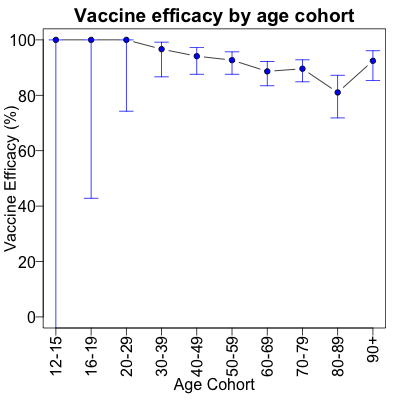 This is a vexing question. The initial Israeli data,
examined skeptically with Simpson’s paradox in mind,
indicates that efficacy is not waning. Even on this crummy little blog that nobody
reads, we can calculate vaccine efficacies and their confidence limits properly, to see
that at least in that dataset, waning was not a happening thing.
This is a vexing question. The initial Israeli data,
examined skeptically with Simpson’s paradox in mind,
indicates that efficacy is not waning. Even on this crummy little blog that nobody
reads, we can calculate vaccine efficacies and their confidence limits properly, to see
that at least in that dataset, waning was not a happening thing.
However, at today’s VRBPAC data saying otherwise was presented by Alroy-Preis & Milo [8], who are between them affiliated with more or less all the great Israeli research institutions. Their credentials are as solid as their data is sobering.
 After showing how bad the previous waves were in Israel (hint: very, very bad), and how
fast they got vaccinated (hint: very, very fast), they looked at epidemiology and
breakthrough infections.
After showing how bad the previous waves were in Israel (hint: very, very bad), and how
fast they got vaccinated (hint: very, very fast), they looked at epidemiology and
breakthrough infections.
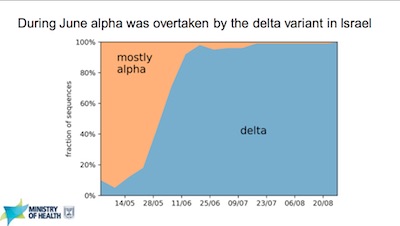
- Slide 8 of their presentation shows that the only strain that matters is Delta, and it’s just plain nasty.
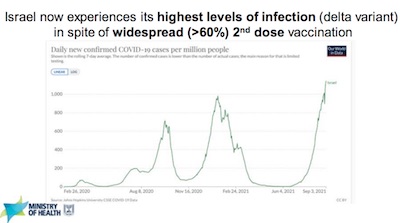
- Slide 9 shows they’re in the midst of a nasty 3rd wave, worse than the others, of all
Delta infections even with > 60% of the population having received 2 doses.
- A couple thoughts from your humble Weekend Editor: Given that Delta seems to have $R_0 \sim 6.7$, that would imply herd immunity at about 85% of the population having immunity (vaccination or previous infection). So 60% just won’t do it.
- So if less than 25% of unvaccinated Israelis have had COVID-19, this is not out of line and may not reflect vaccine efficacy waning, unless it’s concentrated among the vaccinated (as opposed to the unvaccinated, where we more or less expect it to be).
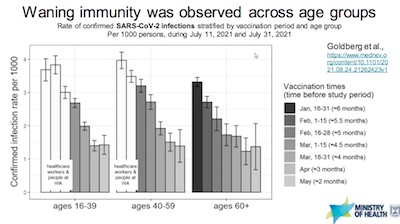
- They’ve addressed some of my concerns about Simpson’s paradox by stratifying by age
(correlated with both time of vaccination and likelihood of hospitalization). Slide 12
shows confirmed infections per 1000 people, across 3 broad age groups. The bars are
colored by when they were vaccinated, showing higher infection rates for those
vaccinated longer, after controlling for age.
- Still, your humble Weekend Editor wishes to point out that those vaccinated earlier were the oldest of the old and the frailest of the frail with complicating pre-existing conditions. You’d expect more breakthroughs there, right?
- So while these data are sobering, I’m not utterly convinced they’ve removed the confounding variables that lead to Simpson’s paradox. Maybe I’m just a skeptical grizzled old statistician. (Well, that’s literally true. The question is whether that’s relevant?)
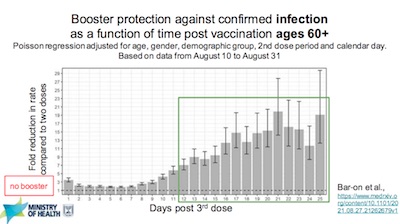
- Whatever reservations I may have, they do not share them. Israel went about giving booster vaccinations with admirable enthusiasm. They did it for their elders first, which makes sense, and the 60+ cohort is now mostly triple-vaccinated. Slide 22 shows the result: a dramatic fold reduction in confirmed infection in that most triple-vaccinated cohort.
- Similarly, a booster gave 10-fold reduction in risk of severe disease, not just infection in the 60+ cohort. That’s good, because severe disease, hospitalization, and death are the things we care about most, not a mild breakthrough infection.
- Best of all, the side effects looked comparable to, or even less than, what was seen in the first 2 doses. They saw only 1 case of myocarditis. (But that’s a problem in younger males, and they vaccinated their elderly first, so the low number of events seen here may not mean much.)
We can argue about the need for boosters, but it’s inarguably true that it worked.
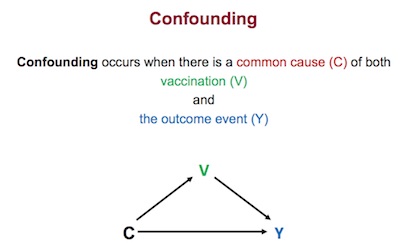
 The counterpoint was presented by Jonathan Sterne, of the University of Bristol, on the
various confounders [9], much like age was the confounder of
vaccination and hospitalization in
the Simpson’s paradox example.
The counterpoint was presented by Jonathan Sterne, of the University of Bristol, on the
various confounders [9], much like age was the confounder of
vaccination and hospitalization in
the Simpson’s paradox example.
- On slide 13, he points to 13 studies, examining in-the-wild vaccine efficacy in multiple countries where Delta is the dominant strain. Even the 95% lower confidence limit on vaccine efficacy is still usually > 80% (with a few exceptions).
- On slide 17, he has a beautifully clear graphic, reproduced here, explaining how confounders work, like age was a confounder of vaccination status and hospitalization in Israel, leading to the Simpson’s paradox mislead of waning vaccine efficacy.
- He then goes through slide after slide of variables that affect people’s chances of being vaccinated, each of which are plausible or actually known to affect medical outcomes (age, BMI, ethnicity, COPD, dementia, …). Since we haven’t controlled for that, we’re in Simpson’s Paradox Town!
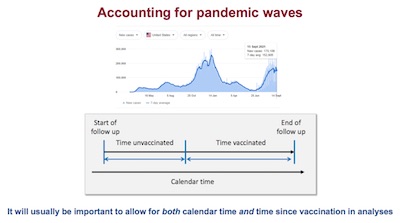
- Very interestingly on slide 39, he points out that the time of vaccination matters in relation to COVID-19 waves. If you spend unvaccinated time during a lull, but get vaccinated right before a wave of infection breaks out, then there’s a problem. The relative risk you’re exposed to has changed: less risk when you were unvaccinated, but more risk after vaccination in the wave of infection. So that’ll make vaccine efficacy look awful, when it’s actually not. (Hey, this guy’s good – I hadn’t come anywhere near thinking of that one!)
- The title of slide 42 sums it up:
The vaccines work brilliantly.
But how should we use observational data to guide policy?
Summary: The Israeli data was quite troubling, but the fact that boosters worked where they were needed (mostly in the elderly) is incontestable. Sterne’s analysis, like our Simpson’s paradox analysis, does contest whether there’s a universal need for boosters.
COVID-19 Epidemiology Update on Delta
 Sara Oliver of the CDC presented an epidemiology update, particularly about Delta in the
US. [10] Our times are very difficult, but the vaccines
continue to work:
Sara Oliver of the CDC presented an epidemiology update, particularly about Delta in the
US. [10] Our times are very difficult, but the vaccines
continue to work:
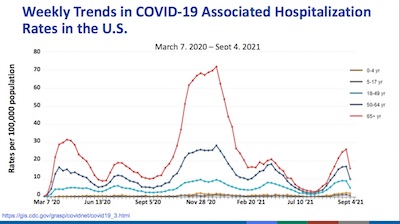
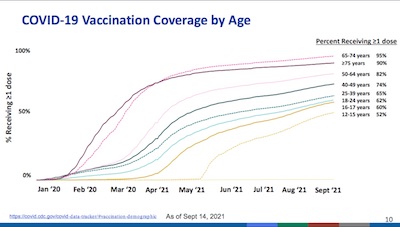
- On slide 6, she shows us the hospitalization rates in the US over time, stratified by age. It shows the recurring wave structure, as we try again and again to be as stupid as possible. Given the high rates for those in their 60s, this is no time to be unvaccinated. It’s dangerous out there.
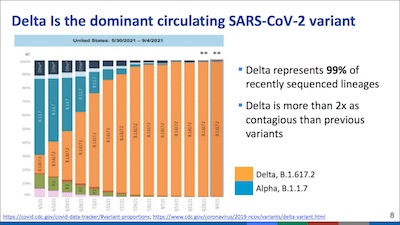
- On slide 8, she shows us that the Delta strain is the only SARS-CoV-2 strain that matters now. It’s just that nasty.
- On slide 12, she shows us the vaccine penetrance in the US over time, stratified by age. To my deep disappointment, it has reached a plateau in all age groups, at levels mostly below herd immunity (say, about 85% for Delta). The only age cohorts that have reached that are the elderly (with almost-ran status for the late middle aged). This should shame all of us in the US.
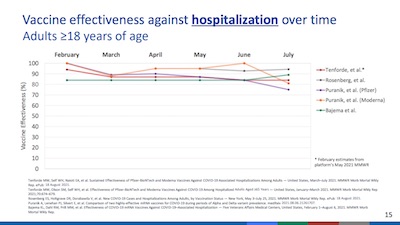
- However, there is some good news about vaccines, which she shows us on slide 15. Vaccine efficacy against hospitalization (not just against mild infection), has remained quite steady over time. There’s barely any change in the last 7 months. The most pessimistic study is still north of 80% efficacy, with the rest around 90%. That’s good news indeed, and pretty strong empirical evidence against vaccine efficacy waning where it matters: severe disease.
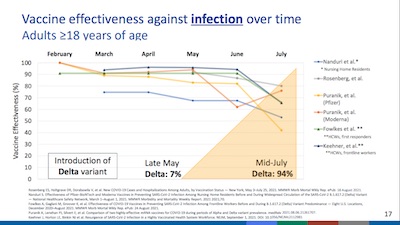
- On slide 17, she examines apparent vaccine efficacy waning against infection (not just severe infection, hospitalization, or death). Looking at the timing, it seems much more likely that this is caused by the introduction of Delta, not by an waning of vaccine efficacy. Not exactly good news (Delta = bad!), but at least the vaccines continue to work where it matters: preventing hospitalization & death.
Summary: While infection, hospitalization, and death rates are high, it is mostly among the unvaccinated. Vaccines continue to provide robust protection against severe disease. Unfortunately, vaccination rates are slowing down, and the defiant few remaining will require mandates to get vaccinated lest they get sick themselves, spread it to others, or – Heaven forbid – evolve yet another variant.
The Question
The VRBPAC will vote on the following question [11]:
Question to the Committee
- Do the safety and effectiveness data from clinical trial C4591001 support approval of a COMIRNATY booster dose administered at least 6 months after completion of the primary series for use in individuals 16 years of age and older?
Please vote Yes or No.
NB: That question implies no stratification by age, which we already know makes huge differences, even on this crummy little blog that nobody reads. Also, going down to age 16 tripped them up a bit in the EUA hearings, getting 4 “No” votes, and in any case the confidence interval on efficacy in 16-17 year olds was ridiculous, as also calculated on this crummy little blog that nobody reads. Also also, the clinical trial for boosters apparently did not include any subjects under 18, so asking for approval for an age group you didn’t test is skating on thin ice.
You’d think Pfizer/BioNTech would have learned from their EUA experience not to propose for low-statistics age cohorts… but here we are.
The Old Reliable(s)
 The livestream video above has the goods in detail. But for a more summary view, we turn
to the formidably reliable Helen Branswell and Matthew Herper at STAT News, who are
apparently live-blogging the affair. [12]
The livestream video above has the goods in detail. But for a more summary view, we turn
to the formidably reliable Helen Branswell and Matthew Herper at STAT News, who are
apparently live-blogging the affair. [12]
Some important introductory points they make are:
- Everybody thinks spacing the doses out further than the initial 3-4 weeks would have resulted in better immunity, but at the cost of slowing down vaccinations. So we chose faster vaccination in the face of a pandemic, but might want to reconsider that in the future.
- Nobody quite knows what Delta (or it’s much-feared hypothetical successor) will do; right now it seems to be moving from the upper airway to the lower airway, which is not news.
- We know a boost will work; the question is whether that’s the best thing to do versus, say, vaccinating everybody else.
- There are other vaccines that require 3 or more doses (Hep B, DTaP, Hib, PVC13, IPV… and that’s just the examples I could find in 30sec!) If the mRNA vaccines for COVID-19 are in that class, “the world needs to know that.”
- Important detail: The VRBPAC’s job is to determine only if treatments meet safety and efficacy needs. It is not about how ot roll out that treatment to the population. So the practical logistics of boosters are off the table for today’s discussion (or supposed to be). Some of that will happen with the CDC’s Advisory Committee on Immunization Practice (ACIP) which meets next Wednesday & Thursday.
A cute detail: Peter Marks, the head of CBER, uses a screen background of a polar bear drinking tea. I might like this guy…
Another “elephant in the room”, as Branswell put it, is the rare instances of myocarditis and pericarditis that sometimes happen in young men, and whether a 3rd dose will raise that frequency. Nobody knows, because the clinical trial here was too small to be powered to see such a rare event.
However, Israel has boosted 1.2 million people and seen exactly 1 case of this sort of heart inflammation. So that’s potentially a good omen if not exactly the RCT statistic we’d like to have in the best of all possible worlds. The acting chair of VRBPAC, Arnold Monto, cautioned that Israel is doing the oldest first, so their data under-represents males under 30 who are most at risk for myocarditis/pericarditis. So… the risk looks low, but really nobody knows.
Another slightly cute detail: Pfizer’s presentation ran long, and when they attempted to review the Israeli data that had already been discussed, the chair cut them off. I admit that as a now-retired pharma research scientist, it’s a gleeful fantasy of mine to be able to tell a senior VP of a pharma company to sit down and shut up. (Perhaps I am a bad person. But it’s hard to resist the cravings for a teensy slice of schadenfreude pie.)
The public comment period was as crazy a mixed bag as one might expect, including fairly crazy skeptics who think all COVID-19 vaccines are unsafe. Le sigh. Also present in the public comment period: Paul Alexander, a former Trump administration official who is skeptical of all the COVID-19 vaccines. He used to yell at FDA & CDC officials, interfere with their reports where they did not meet Trump’s fantasies, and today generally ranted about the story of rapper Nicki Minaj’s cousin’s friend in Trinidad with the swollen testicles… anything except the subject of the meeting, which is booster safety and efficacy. Alexander’s Trumpian boss Michael Caputo apparently accused scientists of “sedition”. So imagine the attention paid to their remarks.
Interestingly, Jonathan Sterne of the University of Bristol presented some data from the UK noting that there are many confounders with vaccine efficacy. We explored some of that in the Israeli data, showing that age caused a strong Simpson paradox effect. Sterne showed a number of similar things happened in the UK, and might mislead one to think the vaccines are fading when they are not. Herper comments that this was done in such a sotto voce manner, that people might not have paid attention. Honestly, do we really have to scream all the time?
At least in Herper’s opinion, the morning failed to resolve definitively 2 important questions that one would have hoped to have answered:
- Is vaccine efficacy waning against hospitalization and death, not just any infection at all?
- Are there safety issues with a 3rd dose, like myocarditis/pericarditis in young males, that would shift the risk vs benefit?
First Vote
At 3:30pm, the vote on the question above came out negative: 3 Yes, 16 No, 0 Abstain. Interestingly, that’s 19 votes out of the 18 committee members! There was one accidental vote, apparently by a speaker; their choice was unclear, but it could not have changed the outcome. (This will be a subject of gossip in the months to come: who barged in and voted without the right?)
Now they’re discussing changing the question, to see if they can endorse boosters for some subgroups of people, if not the entire population. Remember my prediction above, that this would be the sensible outcome? Maybe, just maybe…
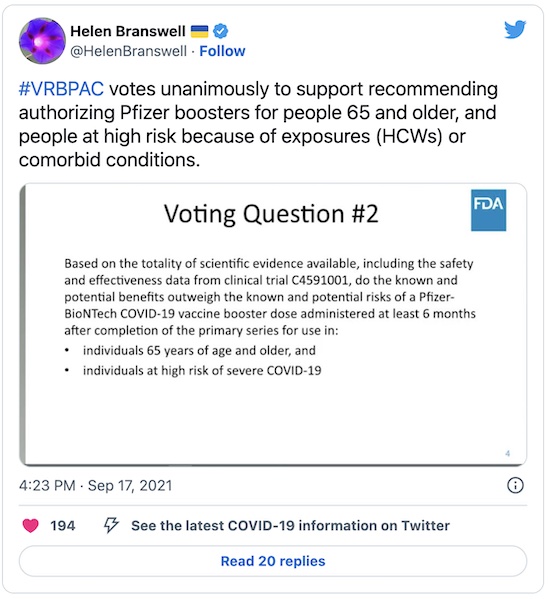
Second Vote
Right, so now they’ve rephrased the question, to be whether there should be boosters for those over 65 and those at high risk of COVID-19 (presumably the immunocompromised and healthcare workers). It passed unanimously: 18 Yes, 0 No, 0 Abstain. (And no “extra” votes, this time.)
This will be a recommendation for an Emergency Use Authorization for boosters in those groups. Your humble Weekend Editor will be eligible in next week if the interval is 6 months from the 2nd dose; the Weekend Editrix alas, is not eligible as she is under 65 (and thus strong in her own right, so that’s not a bad thing).
That seems eminently sensible to me! After all, I predicted it this morning at the top of this post, though I was too cynical to think it would actually happen. Mea maxima culpa.
Next, the ACIP committe of the CDC meets for 2 days next week to figure out what to do with this recommendation. People who got Moderna or J&J have yet to get a decision.
The Weekend Conclusion
 Hunh. Lookitthat. They went and did The Sensible Thing?!
Hunh. Lookitthat. They went and did The Sensible Thing?!
Apparently I am a cynical old grouch. (Like you didn’t know that already.)
I look forward to my booster dose soon, since 6 months after my second dose will be exactly 1 week from today!
Addendum 2021-Sep-21: ZDoggMD on the FDA VRBPAC Booster Hearing
For those who style themselves ‘visual learners’, here’s a video by ZDoggMD (aka Dr. Zubin Damania) talking about the FDA booster hearing. Keep in mind that he’s an entertainer, so he has to be a little ranty and over-the-top to get YouTube views. However, he’s a UCSF-trained doctor, spent 10 years as a hospitalist at Stanford, etc. – guy knows how the medical system works (and doesn’t work).
It sounds like he and I are mostly aligned, in that the evidence of the need for boosters is a little shaky:
- the Israeli data is full of confounders like age (and Simpson paradox effects),
- the Pfizer doses were only 3 weeks apart which may not have been enough (though it got people vaccinated faster, so there’s that trade-off),
- the efficacy vs severe disease or hospitalization or death is still like 90%,
- while boosters boost antibodies but those aren’t the only measure of immune competence,
… and so on. So boosters make sense for those most at risk: elderly, health care workers, and other high-risk situations (details to be worked out at the CDC’s ACIP meeting).
Where we differ is mandates.
He skews more libertarian and wants to try more to persuade the vaccine defiant. I think we’ve done that for almost a year now; I’ve totally lost patience with them and favor mandates. My attitude is hey, if guys like Zubin here can make persuasion work at a scale to make a dent in the unvaccinated population, then great, they should do that. But the empirical result is that it hasn’t worked: the vaccine defiant just shrug it off, spit another conspiracy theory at you, and hork down some horse paste. Time to try something more effective.
Perhaps Zubin is a nicer man than me. I just want us all to be vaccinated, except for rare medical exceptions where that’s not possible.
Addendum 2021-Sep-22: FDA Approves Pfizer Boosters
 Helen Branswell at STAT News is now reporting [13] that
the FDA has today officially authorized Pfizer’s COVID-19 booster for those over 65 or at
high risk. They don’t have to follow the VRBPAC recommendation, but usually do; this
situation turns out to be part of the “usual”.
Helen Branswell at STAT News is now reporting [13] that
the FDA has today officially authorized Pfizer’s COVID-19 booster for those over 65 or at
high risk. They don’t have to follow the VRBPAC recommendation, but usually do; this
situation turns out to be part of the “usual”.
Per the CDC, there are about 53 million Americans over 65, and 17-20 million are health care workers (no word on the overlap). Possibly 100 million have complicating conditions. That adds up to about half the US population, so there’s going to be some arguing about what exactly constitutes “high risk”. They’re considering health care workers, teachers, daycare staff, grocery workers, people in homeless shelters or prisons… all kinds of people.
The ACIP meets tomorrow and will vote on whether to advice the CDC to recommend boosters, and for whom. The CDC will probably accept that recommendation, but again no guarantees.
Addendum 2021-Sep-23: CDC ACIP Meeting Approves Most Boosters
 I didn’t go over the materials in detail, since I’m taking a day off to do other things. However,
the meeting presentations are here,
for your perusal.
I didn’t go over the materials in detail, since I’m taking a day off to do other things. However,
the meeting presentations are here,
for your perusal.
Also, the redoubtable Helen Branswell has a Twitter thread
that more or less sums everything up for
yesterday’s meeting
and today’s meeting.
Her story is up at STAT News. [14]
Like the FDA VRBPAC, the CDC ACIP has re-engineered the question into 4 pieces. Should we give the Pfizer/BioNTech 3rd shot booster to:
- Age > 65yr or LTCF residents? Passed: 15-0.
- Age 50-64yr with underlying conditions? Passed: 13-2.
- Age 18-49yr with underlying conditions? Passed: 9-6.
- Age 18-64yr in occupational/institutional high risk setting? Failed: 6-9
You can see the unanimity degrades as the booster group expands.
Addendum 2021-Sep-24: Another Twist, Another Turn
 Today the CDC issued its official guidance. [15]
Interestingly, it overruled the ACIP committee and said categories 1 & 2 should
get boosters, while categories 3 & 4 may get boosters. In saying so, they’ve
disagreed with the ACIP verdict of “no” in category 4.
Today the CDC issued its official guidance. [15]
Interestingly, it overruled the ACIP committee and said categories 1 & 2 should
get boosters, while categories 3 & 4 may get boosters. In saying so, they’ve
disagreed with the ACIP verdict of “no” in category 4.
So basically they widened the categories of people to get boosters beyond what the ACIP recommended.
I don’t quite know how to feel about this:
- On the one hand, I think the risk of boosters is very low and the benefit is at least medium if you look across the whole population, so that’s probably overall favorable.
- On the other hand, I don’t like the agencies overruling their outside expert advisory committees, and I’d like to see us putting more effort getting first doses into the vaccine resistant.
So I guess I feel… a bit of both ways, maybe?
Gonna get a booster myself, though. You should too, if you’re eligible.
By my calculation above, I think we’re up to a bit over half the US population being eligible, so it’s not a stretch that maybe you’re probably eligible. Here’s the list of qualifying medical conditions, and it’s pretty broad. Even being overweight but not obese, i.e., $25 \le \mathrm{BMI} \le 30 \mathrm{kg}/\mathrm{m}^2$, will do. (Personal point of reference: my BMI after 1.5 years of staying at home in a pandemic is 28.9. If you’ve gained a few pounds in the last year and a half, you can put those pounds to ironic use and get a booster. Make those pounds pay rent; you can lose weight afterwards.)
Strange times we live in. Strange times. [16]
Notes & References
1: FDA, “FDA to Hold Advisory Committee Meeting to Discuss Pfizer-BioNTech’s Application for COVID-19 Booster”, FDA Office of Media Affairs, 2021-Sep-01. ↩
2: FDA & CBER, “167th Meeting of the Vaccines and Related Biological Products Advisory Committee September 17, 2021 Agenda”, US FDA Media releases, 2021-Sep-17. ↩
3: Pfizer/BioNTech, “BNT162b2 [COMIRNATY (COVID-19 Vaccine, mRNA)] Evaluation of a Booster Dose (Third Dose) VACCINES AND RELATED BIOLOGICAL PRODUCTS ADVISORY COMMITTEE BRIEFING DOCUMENT”, www.FDA.gov, timestamped 2021-Sep-14. 53pp. ↩
4: Pfizer Media Relations & BioNTech Media Relations, “Pfizer and BioNTech Initiate Rolling Submission of Supplemental Biologics License Application to U.S. FDA for Booster Dose of Comirnaty® in Individuals 16 and Older”, Pfizer Press Releases, 2021-Aug-25.↩
5: FDA Staff, “FDA Briefing Document Application for licensure of a booster dose for COMIRNATY (COVID-19 Vaccine, mRNA)”, www.FDA.gov, timestamped 2021-Sep-15. 23pp. ↩
6: D Boyce & WC Gruber, “BNT162b2 [COMIRNATY® (COVID-19 Vaccine, mRNA)] Booster (Third) Dose”, Pfizer slides for VRBPAC 2021-Sep-17 meeting, 2021-Sep-17. ↩
7: J Lee, “FDA Review of Effectiveness and Safety of COMIRNATY (COVID-19 Vaccine, mRNA) Booster Dose Biologics License Application Supplement”, FDA slides for VRBPAC 2021-Sep-17 meeting, 2021-Sep-17. ↩
8: S Alroy-Preis & R Milo, “Booster protection against confirmed infections and severe disease - data from Israel”, Israeli Ministry of Health, Weizmann Institute, Technion, Gertner Institute, and Hebrew University slides for VRBPAC 2021-Sep-17 meeting, 2021-Sep-17. ↩
9: J Sterne, “Real-world effectiveness of COVID-19 vaccines”, University of Bristol slides for VRBPAC 2021-Sep-17 meeting, 2021-Sep-17. ↩
10: S Oliver, “Updates to COVID-19 Epidemiology and COVID-19 Vaccines”, Centers for Disease Control and Prevention slides for VRBPAC 2021-Sep-17 meeting, 2021-Sep-17. ↩
11: FDA Staff, “Question for the Committee”, FDA VRBPAC 2021-Sep-17 meeting materials, 2021-Sep-17. ↩
12: H Branswell & M Herper, “Tracking the FDA advisory panel meeting on Covid-19 booster shots”, STAT News, 2021-Sep-17. ↩
13: H Branswell, “FDA authorizes Pfizer’s Covid-19 booster for people over 65 or at high risk”, STAT News, 2021-Sep-22. ↩
14: H Branswell, “Advisory committee recommends wide swath of Americans be offered Covid-19 vaccine boosters”, STAT News, 2021-Sep-23. ↩
15: CDC Media Relations, “CDC Statement on ACIP Booster Recommendations”, CDC Newsroom Releases, 2021-Sep-15. ↩
16: And yes, you’re perfectly within your rights to tell me I sound like an old man when I say stuff like that. Doesn’t make the times any less strange, though.↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.