Pfizer/BioNTech vaccine passes FDA external advisory committee!
Tagged:COVID
/
PharmaAndBiotech
/
Politics
Today is a good day: the FDA’s external advisory committee passed the Pfizer/BioNTech COVID-19 vaccine at 6:30pm EST.
VRBPAC committee meeting
 STAT News reports as of now [1]
that at 6:30pm, after a day of robust discussion, the FDA’s external advisory committe
gave thumbs-up to the Pfizer/BioNTech COVID-19 vaccine. The relevant committee in this
case is the Vaccines and Related Biological Products Advisory Committee (VRBPAC). The
vote was 17-4-1, i.e., overwhelmingly in favor, with one curious abstention.
STAT News reports as of now [1]
that at 6:30pm, after a day of robust discussion, the FDA’s external advisory committe
gave thumbs-up to the Pfizer/BioNTech COVID-19 vaccine. The relevant committee in this
case is the Vaccines and Related Biological Products Advisory Committee (VRBPAC). The
vote was 17-4-1, i.e., overwhelmingly in favor, with one curious abstention.
The main question, and 2 discussion questions, posed to the committe by the FDA were:
- Based on the totality of scientific evidence available, do the benefits of the Pfizer-BioNTech COVID-19 Vaccine outweigh its risks for use in individuals 16 years of age and older? (NB: usually that would be 18 or older, which caused some discussion.)
- Please discuss any gaps in plans described today and in the briefing documents for further evaluation of vaccine safety and effectiveness in populations who receive the Pfizer-BioNTech Vaccine under an EUA.
- Pfizer has proposed a plan for continuation of blinded, placebo-controlled follow-up in ongoing trials if the vaccine were made available under EUA. Please discuss Pfizer’s plan, including how loss of blinded, placebo-controlled follow-up in ongoing trials should be addressed. (NB: this caused some discussion about the ethics of giving the placebo subjects the vaccine, which would shut off follow-up.)
This committee approval is not the final step; the committee’s report is a recommendation to the FDA, not a binding decision. There are times in the past when the FDA has chosen to disregard the opinion of external scientists & medical pracitioners, but it’s pretty rare. It’s highly likely that the FDA will issue very soon a formal EUA for the Pfizer/BioNTech COVID-19 vaccine. [2]
It may take them a couple days to work through all the paperwork, get the authorizations into the FDA computers, and so on – but in a situation like this lots of people would be happy to work through the weekend if it meant vaccinations could start on Monday.
So here at Chez Weekend, we’re betting on Monday.
Formal scientific publication in the NEJM
 Eric Rubin, a VRBPAC member and an immunologist at Harvard’s HT Chan School of Public
Health, co-wrote an (audio interview) editorial in the New England Journal of Medicine this
morning [3] saying it was “impressive enough to hold up in
any conceivable analysis” (quite a strong endorsement, thinks this grizzled old
statistian) and: “This is a triumph.”
Eric Rubin, a VRBPAC member and an immunologist at Harvard’s HT Chan School of Public
Health, co-wrote an (audio interview) editorial in the New England Journal of Medicine this
morning [3] saying it was “impressive enough to hold up in
any conceivable analysis” (quite a strong endorsement, thinks this grizzled old
statistian) and: “This is a triumph.”
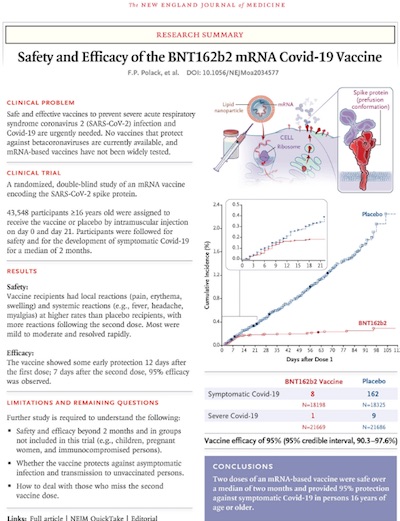
Also, the Phase 3 trial report on clinical trial NCT04368728 made it past peer review and was published this morning, also in the prestigious NEJM. [4]
It’s a thing of beauty. I do not say that lightly.
Issues that concerned VRBPAC members
Pediatric patients
Apparently, there was discussion of including 16-17 year olds in the vaccination population, instead of the usual 18+ age limit. This triggered some discussion that the vaccine has not had a pediatric trial (fair enough), nor has it had an early pediatric trial (infants & kids), nor on pregnant or lactating women (so-called “DART studies”, for “developmental and reproductive toxicity studies”), and so on. All fair enough, but not exactly on target for a vaccine for everybody else, especially when apparently pediatric and pregnant trials are planned already.
The pregnancy issue is a real one: health care workers are justifiably at the front of the line for vaccination, 75% are women, and many are of child-bearing age. There could be, according to the CDC, about 330,000 health care workers pregant or lactating.
The trial so far has had 153 participants ages 16-17, so we’re not blind. Young people down to age 12 have been vaccinated for a follow-on study, but those weren’t in the EUA.
Apparently, some of the “no” votes were from members who wanted to exclude anybody under 18 from getting the vaccine in advance of pediatric trials, in an abundance of caution.
Crossover design
The commitee wanted to get the subjects in the control arm vaccinated as soon as possible (see “crossover design of clinical trials”). Pfizer pointed out that this would be impractical: 44,000 patients would have to come for 2 more appointments, sign completely new consent forms, and might even refuse (meaning there would be no follow-up data from them).
Basically, it would mess up the trial, and they are going to be vaccinated soon with the rest of the popuation anyway.
Severe allergies
Some people just have severe reactions to vaccines. If we know that about a patient, then they should be required to site around for 45min or so under observation by a nurse with an epi-pen, or something like that.
Pfizer did happily note, though, that they had only 1 severe COVID-19 case (due to brief drop in blood O2 levels) in the treatment arm.
Conspiracy theories
There were, of course, public comments. And some of them were wild and wooly accusations that vaccines permanently injure people (sigh), or the extreme canard that they contain aborted fetal cells (wildly false-to-fact), or bizarre assertions that the clinical trial enrolled only whites (it was carefully designed to have multiple ethnic groups and both genders adequately represented: 26% Latino, 10% Black).
Why do we have to be so crazy? Given we have to be so crazy, why do we also have to be so damn stupid?!
Conclusion: today is a good day
It’s highly likely vaccinations can begin in earnest next week. Moderna is following fast, and will likely approximately double our vaccination capability by the week after next.
Though I still don’t understand who on the VRBPAC abstained, or why… (Later: it appears most of the “no” votes as well as the abstention were over the issue of 16-17 year olds vs age 18 and over. The younger ones were added later in the trial, and there’s less safety data for them. That will be cleared up next year with pediatric trials.)
Addendum 2020-Dec-11: HHS Secretary Azar says FDA informed Pfizer it will grant EUA
Appearing on Good Morning America with George Stephanopoulos early this morning, HHS Secretary Azar confirms that the FDA has informed Pfizer this morning that it has granted the Emergency Use Authorization. Pending a bit of paperwork, vaccinations will begin Monday or Tuesday of next week.
Told you it would be Monday. :-)
Also reported on CNN [5], WaPo [6], and most importantly by the FDA itself [7], which said in part:
Following yesterday’s positive advisory committee meeting outcome regarding the Pfizer-BioNTech COVID-19 vaccine, the U.S. Food and Drug Administration has informed the sponsor that it will rapidly work toward finalization and issuance of an emergency use authorization. The agency has also notified the U.S. Centers for Disease Control and Prevention and Operation Warp Speed, so they can execute their plans for timely vaccine distribution.
Addendum 2020-Dec-12: the Republican clown car pulls up & excretes Trump
 In one of his trademark acts of performative cruelty & ignorance, Trump intervened in
the FDA approval process, even though it’s finished for all but a bit of paperwork. On
Friday afternoon, he publicly threatened FDA Commissioner Hahn that if he didn’t get the
approval finished by Friday evening, he would have to resign; this for an EUA that was on
track for approval the next day. [8] (Just to set the scale
on your paranoia barometer, Trump is apparentlly asking suspiciously
if Pfizer is run by Democrats who are out to get him. [9])
In one of his trademark acts of performative cruelty & ignorance, Trump intervened in
the FDA approval process, even though it’s finished for all but a bit of paperwork. On
Friday afternoon, he publicly threatened FDA Commissioner Hahn that if he didn’t get the
approval finished by Friday evening, he would have to resign; this for an EUA that was on
track for approval the next day. [8] (Just to set the scale
on your paranoia barometer, Trump is apparentlly asking suspiciously
if Pfizer is run by Democrats who are out to get him. [9])
As a result, instead of being approved Saturday morning, it was approved Friday night. This makes no difference whatsoever in the outcome, or in the timing of vaccinations.
Trump simply had to be seen personally as the cause of the vaccine, even if it involved pointless whip-cracking on the backs of people busily pulling off a medical miracle. Never mind that Pfizer has refused any money from Operation Warp Speed, and thus the Trump cabal had absolutely nothing to do with the result. By this act of performative stupidity and cruelty, his “basket of deplorables” will believe otherwise. That’s what basket cases do, apparently.
Notes & References
1: STAT Staff, “FDA advisory panel endorses Pfizer/BioNTech Covid-19 vaccine”, STAT News, 2020-Dec-10, 6:30pm. ↩
2: Does it even have a name yet? We can’t keep calling it “the Pfizer/BioNTech COVID-19 vaccine”. The formal name for the studies is the BioNTech id, BNT162b2. That’s not exactly an improvement! ↩
3: E Rubin, et al., “Audio Interview: SARS-CoV-2 Vaccination and Vulnerable Populations”, NEJM, 2020-Dec-10.↩
4: C4591001 Clinical Trial Group, “Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine”, NEJM, 2020-Dec-10.↩
5: C Maxouris, J Hanna, N Thomas, “FDA intends to ‘proceed towards an authorization’ of Covid-19 vaccine, official says, as cases soar”, CNN, 2020-Dec-11. ↩
6: L McGinley, C Johnson, J Achenbach, “FDA says it ‘will rapidly work toward’ authorization of Pfizer-BioNTech coronavirus vaccine”, Washington Post, 2020-Dec-11. ↩
7: FDA Commissioner Stephen Hahn, Center for Biologics Evaluation and Research (CBER) Peter Marks, “Biological Products Advisory Committee Meeting”, FDA.gov web site, 2020-Dec-11. ↩
8: K Collins, K Liptak, J Acosta, “White House chief of staff told FDA chief vaccine must be authorized Friday or he needs to resign”, CNN, 2020-Dec-11. ↩
9: A Suebsaeng, S Stein, “Trump Grows Increasingly Angry With FDA, Wonders if COVID Vaccine Makers Are ‘Democrats’”, Daily Beast, 2020-Dec-11. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.