US FDA Grants Emergency Use Authorization for Bivalent Classic/Omicron COVID-19 Vaccines
Tagged:COVID
/
PharmaAndBiotech
Today the FDA granted Emergency Use Authorization (EUA) to both Pfizer and Moderna’s bivalent COVID-19 vaccines for classic and Omicron/BA.4/5 variants.
Bivalent Classic/Omicron Vaccines EUA’d at FDA
The redoubtable Helen Branswell and Matthew Herper of STAT News are on the case. Branswell’s tweet first alerted us:
She’s pointing to Herper’s summary article at STAT News. [1] A little digging, of course, takes us to the original FDA press release. [2], confirming what Branswell & Herper wrote. Also, the PBS News Hour has a longish (42m55s) interview with FDA Commissioner Robert Califf and CBER Director Peter Marks being interviewed by Lauren Gardner, Cheyenne Haslett, Fiona Rutherford, Ferdus Al-Faruque, Alexander Tin, Michael Erman, and Matthew Herper. [3] (I haven’t finished watching it as of this writing; there’s a lot of stuff there!)
Some of the high points:
- The FDA did not convene the VRBPAC, the committee of outside experts, to pass judgment. Likely that’s because, as we’ve previously blogged, the VRBPAC specified by overwhelming vote this exact formulation on 2022-Jun-28. No need for further review; the FDA just did what they previously said they would do.
- Both vaccines are bivalent, i.e., contain mRNA for the spike protein of the classic Wuhan virus as well as the fast-spreading Omicron/BA.4/5 variant. (Thanks to commenter Mike who cut through my post-COVID brain fog to point out in a comment on a previous post that BA.4 and BA.5 variants have high homology in the spike protein, sharing mutations at L452R and F486V.)
- Pfizer/BioNTech is authorized for ages 12 and up. Moderna is for ages 18 and up. No real idea why the difference, other than path-dependence on previous submissions and maybe the fact that Moderna tends to dose higher.
- More time spent indoors during fall & winter in the US is a good reason to enhance COVID-19 immunity, as is the fact that Omicron/BA.4/5 may be the most contagious disease yet encountered by humanity. Omicron/BA.5 now accounts for about 89% of cases in the US, with the bulk of the rest being BA.4. So a vaccine that hits both of those and the classic strain looks pretty much spot on.
- Pfizer says they’ll ship immediately after final QC checks, and they have capacity to ship 15 million doses by 2022-Sep-09. Moderna is also shipping, though I couldn’t find out how many by when. Some sites may get doses as soon as this coming Saturday.
- Some people are complaining that no human clinical trials have been done on this
particular variant of the COVID-19 vaccine. However:
- Previous trials have been done on the Beta variant, the Gamma variant, and both
monovalent and bivalent BA.1. All of them had 3 interesting properties:
- They worked, especially well against the variants targeted, but also against others.
- They were as safe as the original vaccine, i.e., very safe as shown by the original clinical trial and now by more than several billion doses given worldwide.
- The trials took so long that by the time they were done, the virus had moved on to a new variant. So the trial was a tactical mistake.
- New influenza vaccines have a similar problem: very time-sensitive formulations, with a limited window for a trial. Fortunately, we’ve done a lot of flu vaccines in the past, and know how to extrapolate from animal studies without requiring a new human trial every year. That’s the strategy being applied here, and I have high confidence that it is the proper one: be fast, rely on past experience to guide your safety requirements.
- Previous trials have been done on the Beta variant, the Gamma variant, and both
monovalent and bivalent BA.1. All of them had 3 interesting properties:
So: it looks like the bivalent classic/Omicron vaccines are safe (based on monovalent studies and on the bivalent study with BA.1, and based on animal data with the bivalent BA.4/5 version); it also looks like they are massively effective against the Omicron variant. (Unfortunately, I don’t have a Kaplan-Meier curve to show you, but the previous data on classic/BA.1 looked excellent, and we have no reason to think differently here.)
Next Steps
… because of course there’s a next step before this can go into arms, right? The CDC’s Advisory Committee on Immunization Practice (ACIP) has to meet and recommend this FDA-approved therapy for practice in the US. The ACIP meets tomorrow and Friday, to discuss exactly this (see agenda item scheduled for 1pm - 5pm Friday). [4]
So probably we’ll have the final thumbs up (or maybe down, if something disastrous turns up) by tomorrow afternoon.
The Weekend Conclusion
Availability is apparently expected to start in September. We may have to wait a bit, since here at Chez Weekend we’re just now recovering from a lovely late summer case of COVID-19.
But then… vax us up!
Addendum 2022-Aug-31, afternoon
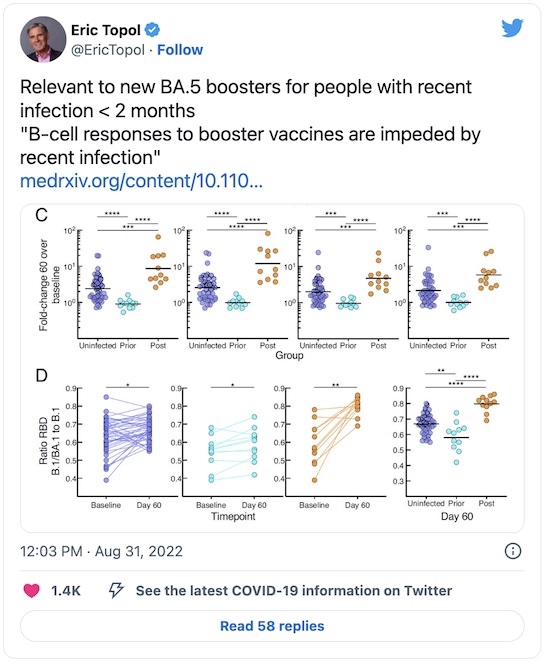
The indispensable Eric Topol points out a preprint that dropped on medRχiv today [5]:
So basically recent (< 2 months) COVID-19 infection impairs B-cell responses induced by the new BA.5 booster.
That means your humble Weekend Editor & Editrix need to wait a bit, as we just finished a regrettable COVID-19 infection. We started testing negative (post-rebound) in late August, so we should get the new vaccine in late October or early November.
One presumes the upcoming CDC guidelines to be decided tomorrow will have summat to say on this subject.
Annoying, but good to know.
Addendum 2022-Sep-01: CDC ACIP Meeting
Our trusty safari guide, Helen Branswell of STAT News, is live-tweeting the meeting of the CDC Advisory Committee on Immunization Practices to set practice recommendations for the bivalent classic/Omicron vaccines:
I’m not gonna trawl through all the primary presentations myself, this time. (Post-COVID-19 brain fog, you know. But if you want to make your own deep dive, then top off your tanks and go here to get the slide decks.) So I’m gonna rely on Helen to be my guide to what’s happening (it took her 62 tweets!). Seems like a good option for my tired brain.
Some interesting highlights from her Twitter thread:
- A few ACIP members did not bother to show up, which is more than a mite peculiar. Initially there was confusion because they didn’t do the roll call in alphabetical order, so it was hard to tell who was there. But after a bit of back-n-forth, it looks like Kevin Ault is the only missing one, so 14/15 voting members are present. Good enough.
- We’ve given 12.6 billion – “billion” with a “b” – doses of COVID-19 vaccines since late 2020. (Yeah, that’s a lot. But: Thre are about 8 billion currently living humans, so to get everybody double-vaxxed and double-boosted would be about 32 billion.)
- In the US, it’s 609 million doses. With a bit over 300 million people, that means lots
of people are unvaxxed, under-vaxxed, or not boosted. This is why COVID-19 won’t go
away.
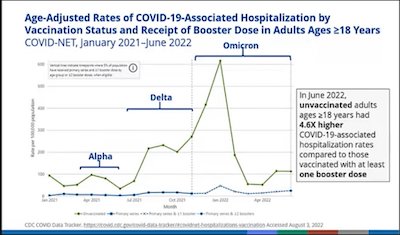
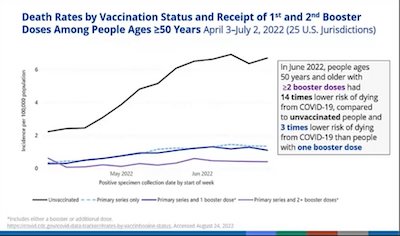
- Here’s a slide from Heather Scobie of the CDC, documenting that the unvaccinated have been, and still are, the bulk of hospitalizations. Also, a second slide from her documenting that fully vaxxed and boosted adults have 14x lower death risk than the unvaxxed, and that a 2nd booster is 3x better than just 1 booster. This is unsurprising to all except the unvaccinated/unboosted.
- Antigenic cartography is a pain in the neck to explain to non-statistics people.
- It’s pretty much like multidimensional scaling, except considerably more fancy-pants. Getting this into biologist and medical brains is harder than learning the actual math. Comme d’habitude.
- But the basic conclusion is that BA.4/5 are really different, both from ancestral
Wuhan and from other Omicron lineages.
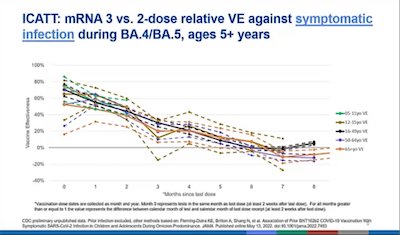
- Here’s a slide from Ruth Link-Gelles showing how rapidly vaccine efficacy wanes, and that this is across all age groups. I must emphasize, very clearly, that this is efficacy against any symptomatic infection. Efficacy against severe disease, hospitalization, and death is much, much better. And that’s what we should care about, not just any old infection. Your humble Weekend Editor got “just any old infection”, and thanks to 4 doses of vaccine + paxlovid, it was merely very annoying.
- Tom Shimabukuro reported on vaccine safety signals, which Helen says he’s done “so often
he could probably do it in his sleep.” While we could all use a nap, what he said
instead of napping was:
- No interesting safety signals went off, including myocarditis in kids being kept at a very low level (like, 0 incidence for little guys 6 months to 4 years, for both vaccines).
- As usual, a little more reactogenicity for Moderna, because of the higher dose.
- Nothing else out of the ordinary.
- They took only 10min for a lunch break, which tells you remote meetings from home can be efficient in unexpected ways. :-) And we’ll just step carefully around the land mines and avoid the public comment period, ok?
- For those concerned about the lack of human trials: (a) this is how we do flu vaccine
updates every year, and (b) Moderna is doing a post hoc clinical trial anyway.
They’ve already done additional trials for Beta, Gamma, and Omicron/BA.1. The trial for
this vaccine concluded enrollment yesterday: $N = 512$ subjects, probably going for
50-200 days, which means we’ll see the results late this year or sometime next year.
(That’s why we use the flu vaccine rules, to get out ahead of the virus!)
- Later: Pfizer, of course, is also doing a similar clinical trial, currently ongoing.
- I’m obscurely pleased to note that Helen was also confused about the spikes of BA.4 and BA.5, as I was a couple days ago. (Hey, it’s not just me and my post-COVID-19 brain fog!) Basically they’re identical spike proteins, so you get to claim a twofer with BA.4/5.
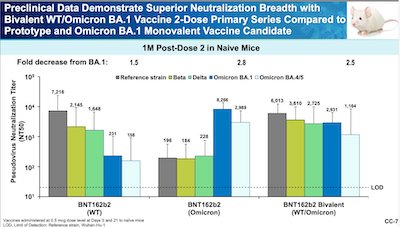
- Moderna claims something quite interesting for their bivalent vaccine, namely that the bivalent vaccine strictly dominates the classic vaccine in terms of antibody levels:
- Here’s a slide from Kena Swanson of Pfizer. It looks at pseudovirus neutralization titers, by fold decrease from BA.1. It compares the classic vaccine, the Omicron/BA.1 vaccine, and the new bivalent classic + Omicron/BA.4-5 vaccine. Result in mice:
- Sara Oliver of the CDC is asking if the ACIP wants to simplify the vaccination series
recommended down to 2 primary shots and 1 bivalent booster. Seems good to me,
effectiveness and simplicity are always in short supply.

- Also, she presented evidence that there was no immune tolerance (ceasing to respond to
later boosters), and – a shown here – little evidence of antigenic original
sin where the immune system gets locked into the original response. Bivalent boosters
seem to improve the situation, if anything.
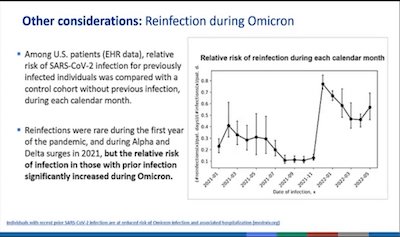
- To illustrate the unmet medical need in the face of Omicron, Oliver presented data on
reinfection of people who’d previously had COVID-19. See the big bump in the curve
starting early 2022? That’s Omicron. That’s why we want an Omicron-specific booster,
to overdamp that wave.
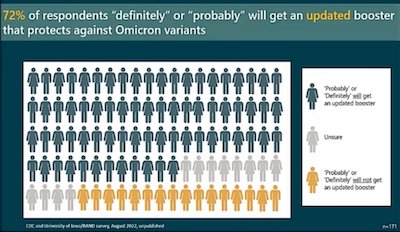
- Vaccine uptake is always an issue, like a toothache that just won’t stop. We
initially had poor uptake of vaccination because of misinformation. Boosters were even
worse, with very few of those eligible in the US getting boosted, despite the evidence
(e.g., above) that boosting dramatically lowers the risk of hospitalization and
death. Why should we believe the bivalent boosters will be any different? As shown
here, the CDC did a survey, finding 72% of subjects would “definitely” or “probably” get
an updated booster, which is way better than the old boosters did. This seems pretty
mysterious in terms of logic, but if it’s true we should pounce on it to get the
bivalent vaccine into people!

- CDC also analyzed the medical benefits, as shown in the slide here.
- The pessimistic left column is if the bivalent booster uptake is about like annual flu shots. The more optimistic right column is if uptake gets to about 80% of the eligible population. Ranges are 95% “credible interval” (the Bayesian version of a confidence limit, and for our purposes more or less the same thing: uncertainty).
- So we’re looking at saving 100k - 160k lives, 1 - 1.7 million hospitalizations, 24 - 48 million infections, and averting $62 - 100 billion dollars in direct medical costs. Looks awfully good to me!
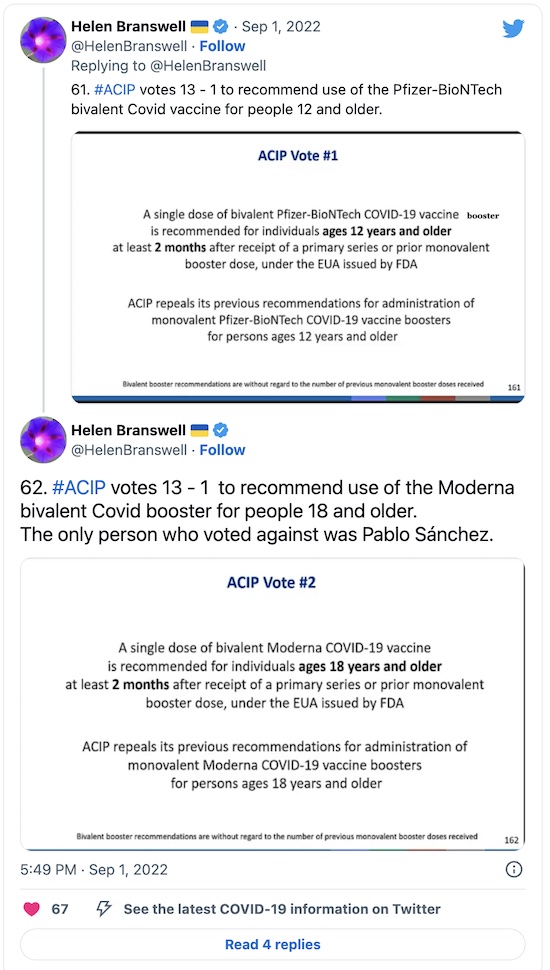
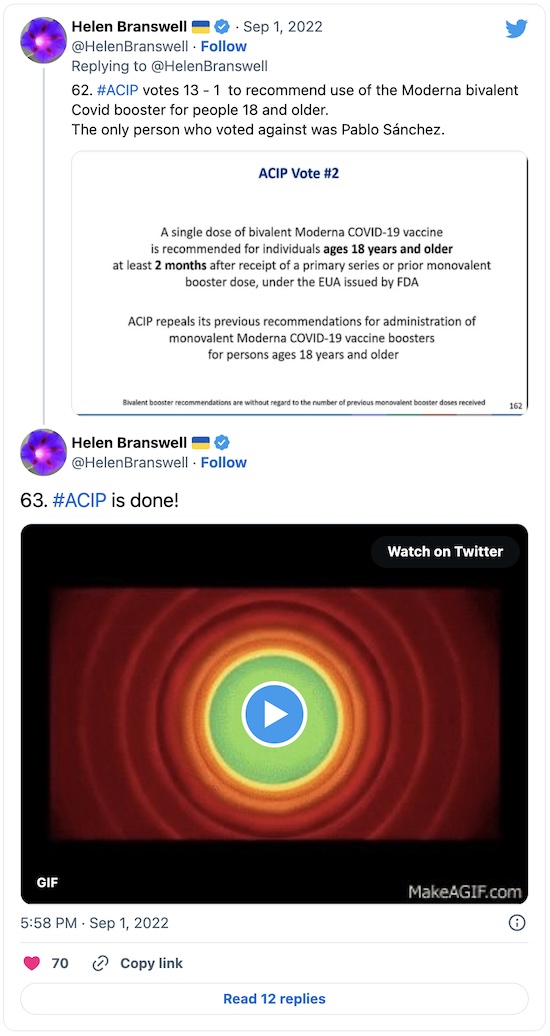
And, that’s it: approval 13-1 with 1 absence, and a reasonable set of practice guidelines for both vaccines:
Honestly, I was really surprised they worried so much about a human trial, in spite of the fact that this very committee handles flu vaccine updates every year in this same way. I just don’t get why that’s not a compelling argument. But the single “No” vote was from Pablo Sánchez, a professor of pediatrics at OSU, who really wanted a clinical trial no matter what the cost of delay. (It would take until at least late November.)
Still, we seem to have converged to the right place.

 Branswell then quickly summarized all this in an article that went up at
STAT News. [6] CDC Director Rochelle Walensky has signed
off on the committee’s recommendation, so it is now A Done Deal. In a weird turn of
events, that happened so fast I can’t find the statement on the CDC web site, though it is
on some news sites. Our safari guides at STAT News are all over
it [7], reporting the CDC Director’s statement faster than
the CDC itself:
Branswell then quickly summarized all this in an article that went up at
STAT News. [6] CDC Director Rochelle Walensky has signed
off on the committee’s recommendation, so it is now A Done Deal. In a weird turn of
events, that happened so fast I can’t find the statement on the CDC web site, though it is
on some news sites. Our safari guides at STAT News are all over
it [7], reporting the CDC Director’s statement faster than
the CDC itself:
On Thursday evening, Centers for Disease Control and Prevention Director Rochelle Walensky signed off on the recommendation made earlier in the day by the Advisory Committee on Immunization Practices, that the newly formulated vaccines be used.
Addendum 2022-Sep-01, later that evening: Official CDC statement
Ah: now the official media statement is up at the CDC web site. [8] It says about what you think it might say:
The following is attributable to Dr. Walensky:
“The updated COVID-19 boosters are formulated to better protect against the most recently circulating COVID-19 variant. They can help restore protection that has waned since previous vaccination and were designed to provide broader protection against newer variants. This recommendation followed a comprehensive scientific evaluation and robust scientific discussion. If you are eligible, there is no bad time to get your COVID-19 booster and I strongly encourage you to receive it.”
The main twist is that they expect “in the coming weeks” to recommend COVID-19 for other pediatric groups, too.
Here at Chez Weekend, we look forward to getting it in late October, once we’re 60 days past our (current) COVID-19 recovery!
Finally…
All my American readers of a certain age were thinking this. But Branswell decided to just go ahead & say the quiet part out loud in tweet #63:
Sums it up.
Notes & References
1: M Herper, “FDA authorizes Pfizer and Moderna Covid boosters targeted against Omicron strains”, STAT News, 2022-Aug-31. ↩
2: A Capobianco, “Coronavirus (COVID-19) Update: FDA Authorizes Moderna, Pfizer-BioNTech Bivalent COVID-19 Vaccines for Use as a Booster Dose”, FDA News Releases, 2022-Aug-31. ↩
3: PBS News Hour Staff, “WATCH LIVE: FDA discusses new guidelines for updated COVID-19 boosters”, PBS News Hour on YouTube, 2022-Aug-31. ↩
4: CDC Staff, “Agenda of ACIP Meeting 2022-Sep-1-2”, US Centers for Disease Control & Prevention, retrieved 2022-Aug-31. ↩
5: C Buckner, et al., “Recent SARS-CoV-2 infection abrogates antibody and B-cell responses to booster vaccination”, medRχiv, 2022-Aug-31. DOI: 10.1101/2022.08.30.22279344. ↩
6: Helen Branswell, “CDC recommends updated Covid-19 boosters”, STAT News, 2022-Sep-01. ↩
7: H Branswell, M Herper, & S Owermohle, “Your questions on the new Covid vaccine boosters answered”, STAT News, 2022-Sep-01. ↩
8: CDC Media Relations, “CDC Recommends the First Updated COVID-19 Booster”, CDC Media Statements, 2022-Sep-01. ↩


Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.