FDA Authorizes Paxlovid
Tagged:COVID
/
PharmaAndBiotech
… aaaaand, today the suspense is over: the FDA has authorized paxlovid.
Paxlovid is legal
 Matthew Herper at STAT News is, as usual, on the case. [1]
There was the official FDA news relase this morning. [2]
Matthew Herper at STAT News is, as usual, on the case. [1]
There was the official FDA news relase this morning. [2]
This is not full approval, but an Emergency Use Authorization. Because… emergency. Another sign of their efforts at speed is that they didn’t convene the AMDAC committee of external advisors to pass judgement, like they did with molnupiravir. This indicates (a) they’re in a hurry to get this out, and (b) the data are really, very, very, good.
The authorization is for age 12 and up.
Mysteriously, it is not authorized for prevention in case of exposure. Maybe the drug interactions of the ritonavir component have people feeling a bit cautious. They did explicitly mention this.
The FDA accepted that the efficacy vs hospitalization was 88%.
The Weekend Conclusion
Here at Chez Weekend, we also approve fully. (Not that anyone should care about that. Instead, care about people getting effective treatment: A Good Thing.)
Addendum 2021-Dec-23: A trawl through the data
One of the drawbacks to sidestepping the advisory committes (AMDAC or VRBPAC) is that we don’t get public availability of the filing documents, the slide decks, and so on. Those are really informative.
But here, as with evusheld, we have to rely on skimpier sources, starting with the FDA press release. So here’s what we’ve got, working forward from the press release and following links:
- The clinical trial itself, called EPIC-HR and given the id NCT04960202 at ClinicalTrials.gov. [3] Sadly, no raw data here to feed into the Weekend R Pipeline. This is one of the reasons I wish they’d had a (fast) AMDAC meeting, because then all the documents would be available.
- The Pfizer fact sheet for patients, parents, and caregivers, i.e., explaining to
non-medical people what’s happening to them. [4] Seems
pretty basic:
- Tell your doc about allergies, liver/kidney disease, pregnancy, breastfeeding, etc.
- Also, I’m glad to see they emphasize telling your doc about all your meds. One of the components of paxlovid is ritonavir, which inhibits liver breakdown via CYP3A of many drugs (deliberately: it makes the other component, nirmatrelvir, last longer in your body). So it interacts by design with other drugs, and you have to watch for that.
- There’s a list on page 3 of drugs that, when taken, mean you can’t have paxlovid. Some are pretty typical, like a couple statins in very wide use; some are odd, like St. John’s Wort (an herbal supplement).
- A worrisome side effect: If you have untreated HIV, paxlovid may lead to some HIV meds not working as well in the future.
- Also, there’s a Pfizer site for consumer side-effect/adverse event reports.
- The Pfizer fact sheet for healthcare providers, with a lot more technical information
and pragmatic use guidelines. [5] This is 29 pages of
detail in the usual medical jargon about how to prescribe it, to whom, what to watch out
for, interactions with other drugs (a lot of them!), cautions around pre-existing
liver and kidney disease, and so on. It goes on for a good long while, and while I at
least glanced at each page, I didn’t give it a deep read.
- There really are a lot of cautions about drug interactions, so it’s good to see that taken seriously.
- The business about HIV drugs not working as well if you already have HIV is explained:
apparently HIV can evolve escape mutations around ritonavir, which is a component of
the usual HIV cocktail. (The HIV cocktail is a complicated combination designed to
make it hard for the HIV virus to evade all of them. But just 1, it can evade.)
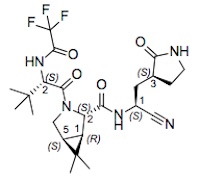
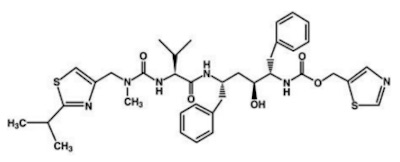
- The former pharma nerd in me can’t resist the structural diagrams for nirmatrelvir and ritonavir. I always admired med chemists: not only can they make these battleship-sized molecules, but they can design a scalable pipeline that does it reliably, over and over, making the exact same thing each time. Repeatably. Verifiably. In a way the correctness of the synthesis can be audited. Amazing.
- There’s also the final, official, all i’s dotted, all t’s crossed report on efficacy.
In the paxlovid arm, $N_p = 1039$ subjects, of whom $K_p = 8$ were hospitalized. In
the placebo arm, $N_0 = 1046$ subjects, of whom $K_0 = 66$ were hospitalized. My
wheezy little R script
says that’s an efficacy against hospitalization of 87.8% (95% CL: 75.1% – 94.0%):
> signif(efficacyAndCL(1039, 8, 1046, 66), digits = 3) LCL Eff UCL 0.751 0.878 0.940 - The same for death rates: 0 in the paxlovid arm, 12 in the placebo arm. That gives an
efficacy vs death of 100% (95% CL: 67.9% – 100%):
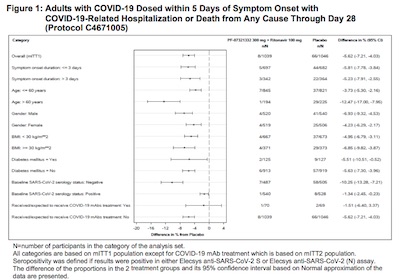
> signif(efficacyAndCL(1039, 0, 1046, 12), digits = 3) LCL Eff UCL 0.679 1.000 1.000 - There’s also (p. 27, click to embiggen) a nice stratification by age, gender, symptom onset delay, BMI, diabetes, and so on of factors thought to be influential. It’s nice to see the % change in hospitalization is statistially significant with nearly all subgroups.
- The FDA’s FAQ on the paxlovid EUA. [6] Basically explanations of what an EUA means, who can prescribe, and so on.
- The official letter from the FDA’s acting chief scientist, granting the
EUA. [7] This one goes on for 11 pages; I’m always
surprised at how long they are.
- Some of it is detail about which sections of the FDA Act apply. If you care about that, then you’re probably a lawyer and don’t need me to tell you about it. If you care about it and are not a lawyer, then I slightly fear you.
- It specifies the prescribing guidelines, the appearance of the pills, the required package inserts, reporting adverse events, monthly reporting on viral variants relevant to resistance (presumably 3CLpro mutants?)… all kinds of stuff.
- The crucial bit:
Having concluded that the criteria for issuance of this authorization under Section 564(c) of the Act are met, I am authorizing the emergency use of PAXLOVID for the treatment of mild-tomoderate COVID-19 in certain adults and pediatric patients, as described in the Scope of Authorization section of this letter (Section II) and subject to the terms of this authorization.
So, no big surprises. Other than that it got done so suddenly, without an advisory committee meeting.
Addendum 2021-Dec-23: And in related news… molnupiravir is also legal
Honestly, I though Merck’s molnupiravir was dead in the water: equivocal recommendation from the AMDAC, worries about mutagenicity particlarly in pregnancy, worries about breeding new variants, weeks of inaction by FDA administrators after the AMDAC meeting, hints that an EUA could be withdrawn if “something better” showed up (like paxlovid)… the list goes on.
But I’m happy to report being wrong: today the FDA gave an EUA for molnupiravir, too. [8] Now, true, they loaded it up with a bunch of restrictions, but those seem appropriate. Such as, “only when other authorized therapies are not accessible or not clinically appropriate”, i.e., last-resort situations.
The pregnancy issue is an interesting one. Women are recommended to get a negative pregnancy test before treatment, then to use contraception during treatment and for 4 days after. Men, on the other hand, are urged to use contraception during treatement and for the following 3 months!
Availability: Merck has hundreds of thousands of courses available now, with 1 million courses over the next few weeks in the US and 10 million to be shipped worldwide. It will be in greater supply than paxlovid for the first few months of 2022.
Now if only somebody would do a combination trial involving paxlovid, molnupiravir, and fluvoxamine. Heck, maybe even monoclonal abs, too. Gotta be a good Loewe synergy score in there somewhere, with all those different mechanisms of action.
Of course you should still get vaccinated.
Notes & References
1: M Herper, “FDA authorizes Pfizer pill to treat Covid-19 in patients as young as 12”, STAT News, 2021-Dec-22. ↩
2: C Tantibanchachai, “Coronavirus (COVID-19) Update: FDA Authorizes First Oral Antiviral for Treatment of COVID-19”, FDA News Releases, 2021-Dec-22. ↩
3: Pfizer Staff, “EPIC-HR: Study of Oral PF-07321332/Ritonavir Compared With Placebo in Nonhospitalized High Risk Adults With COVID-19”, ClinicalTrials.gov, downloaded 2021-Dec-23. ↩
4: Pfizer Staff, “FACT SHEET FOR PATIENTS, PARENTS, AND CAREGIVERS: EMERGENCY USE AUTHORIZATION (EUA) OF PAXLOVID FOR CORONAVIRUS DISEASE 19 (COVID-19)”, FDA.gov, downloaded 2021-Dec-23. ↩
5: Pfizer Staff, “FACT SHEET FOR HEALTHCARE PROVIDERS: EMERGENCY USE AUTHORIZATION FOR PAXLOVID(TM)”, FDA.gov, downloaded 2021-Dec-23. ↩
6: FDA Staff, “Frequently Asked Questions on the Emergency Use Authorization for Paxlovid for Treatment of COVID-19”, FDA.gov, downloaded 2021-Dec-23. ↩
7: JA O’Shaughnessy (Acting Chief Scientist, FDA), “Official Letter of Emergency Use Authorization”, FDA.gov, downloaded 2021-Dec-23. ↩
8: M Herper, “FDA authorizes Merck’s Covid-19 pill, but stresses its use should be limited”, STAT News, 2021-Dec-23. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.