FDA AMDAC Considers Merck's Molnupiravir
Tagged:COVID
/
MathInTheNews
/
PharmaAndBiotech
/
Statistics
Today the FDA’s Antimicrobial Drugs Advisory Committee (AMDAC) meets to decide whether or not to recommend emergency use authorization (EUA) of Merck’s molnupiravir, and antiviral therapy for mild to moderate COVID-19 in adults. Wanna read along?
What’s the sitch?
Merck’s drug candidate for early treatment of COVID-19, molnupiravir, will be considered for EUA (or even approval?) by the FDA’s Antimicrobial Drugs Advisory Committee (AMDAC). This is the first step in a 4-step process, involving:
- The FDA’s AMDAC committee of outside experts has to decide if the risk/benefit is favorable and recommend EUA to the FDA.
- The FDA administration has to say, “Yeah, right: let’s do that.” This is typically a couple days to a week after the AMDAC meeting, though lately with COVID-19 it’s been the evening of the same day.
- The advisory committee of outside experts at the CDC has to recommend to the CDC that this should be recommended medical practice in the US. I don’t know when (or if?) this is scheduled.
- The CDC management has to say, “Ok, fine: that’s now the standard.”
Normally this takes months, maybe a year because people are really careful about stuff on which people will literally bet their lives. Lately, with the urgency of COVID-19 and this being an emergency use authorization and all, it’s instead been taking weeks. So this has been quite fast: somebody is frantically turning the crank really hard on machinery that was never meant to go this fast. So when they get things done, we should applaud them.
If everything goes smoothly, molnupiravir can be manufactured, shipped, stockpiled in pharmacies, and prescribed starting sometime in December. We would, of course, like to see even faster action on Pfizer’s COVID-19 therapeutic, paxlovid.
Today is step 1.
The primary data sources
The FDA announcement of the AMDAC meeting [1] has pointers to all the meeting’s submitted documents and presentations that the AMDAC will consider. For completists and obsessives, all 7 or 8 hours of the hearings are livestreamed, and can be watched on YouTube as seen here.
The agenda for the meeeting [2] is basically pretty straightforward as these things go:
- the usual introductory remarks & boilerplate,
- the Merck presentations,
- the FDA presentations,
- clarifying questions,
- lunch,
- public comment (which I’ll avoid for the sake of my own sanity),
- committee discussions, and
- voting.
The questions and the vote placed before the AMDAC [3] are:
DISCUSSION: Please discuss the potential use of molnupiravir during pregnancy – both in terms of risk and benefit.
a. Comment if you think molnupiravir should be accessible for use in pregnancy in certain scenarios, and if so, please describe what those scenarios might be.
b. Do the concerns regarding the use of molnupiravir during pregnancy extend to the use of molnupiravir in individuals of childbearing potential? If so, are there mitigation strategies that should be considered?DISCUSSION: Please discuss the concern regarding the observed increased rate of viral mutations involving the spike protein among participants receiving molnupiravir. In your discussion, please comment on what, if any, additional risk mitigation strategies or limitations on the authorized population could be considered. What monitoring strategies should be considered to better understand and mitigate these concerns?
VOTE: Do the known and potential benefits of molnupiravir outweigh the known and potential risks of molnupiravir when used for the treatment of mild-moderate COVID-19 in adult patients who are within 5 days of symptom onset and are at high risk of severe COVID19, including hospitalization or death?
a. If yes, please describe the appropriate authorized population such as risk factors for disease progression and pregnant individuals. Please comment on the proposed risk mitigation strategies and if additional risk mitigation strategies are needed.
b. If no, please describe your reasons for concluding that the overall benefit-risk for molnupiravir is not favorable for any population based on the data available at this time.
There are numerous documents to review here:




- Merck submitted a deck of 80 slides to use in its presentation [4], which will be most of the basis for discussion at today’s AMDAC.
- Merck also submitted the formal briefing document of 68 pages & an addendum of 7 pages [5], which are the traditional formal way to present a case to the FDA AMDAC. The committee members are expected to have read this ahead of time.
- As is traditional in these matters, the FDA reanalyzed the data and came to its own conclusions, which will be compared with Merck’s conclusions. The FDA scientists submitted their own slide deck of 96 slides [6] (which includes the FDA introductory remarks for the meeting, as well as extensive toxicology studies).
- There is also FDA’s formal briefing document (44 pages), addendum (3 pages), and erratum (1 page) [7] that are the FDA’s own version of what they think Merck should have said in their formal submission document.
The general idea is to compare the assertion’s of Merck’s scientists with those of the FDA and see if they’re broadly the same. If so, then the results are compared against the rules for EUA (or approval), and after some discussion, there’s a vote over what actions to recommend to the FDA administration.
Our trusty safari guides?
Hmm… normally Helen Branswell and Mattew Herper of STAT News live-blog these things, and we use them as our trusty safari guides through the FDA/CDC jungle. A quick trawl through STAT News comes up empty, though. Maybe we’re on our own today!
Absent our usual safari guides, let’s just take a look through the slide presentations ourselves.
The Merck presentation
- The drug is administered 2ce per day for 5 days at a dose of 800mg (in the form of 4
capsules of 200mg each, so the dose can be titrated down, perhaps?).
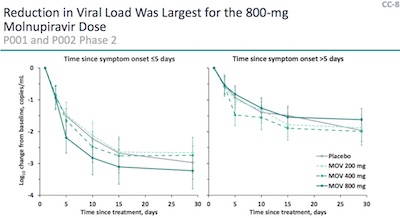
- Treatment must start within 5 days from symptoms. That’s empirically important, as
shown here. The reduction in viral load over time is dramatic for intervention before
5 days past symptoms (left), but it is an order of magnitude weaker for intervention
after 5 days of symptoms (right). That means we need to have a testing system which
is widely available, cheap or free, and fast!
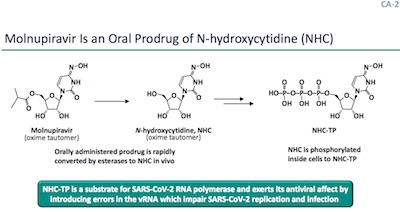
- Interestingly, it’s a prodrug: your liver metabolizes it into the active form, then a
kinse phosphorylates it 3 times, and it’s ready to go. When it gets incorporated into
viral RNA, it can pair with either guanine or adenosine, resulting in many transition
errors in the viral RNA.
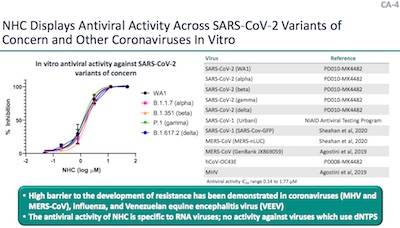
- Very gratifyingly, they did in vitro tests against a number of variants including
Alpha, Beta, Gamma, Delta, and others shown here. The dose-response curve is
encouraging: all variants reached 100% inhibition (good efficacy), and the EC50 for
all looks to be around 2-3μM (good potency). This means that, whatever the
variants are selecting for, it’s unlikely to be conveying immunity to molnupiravir.
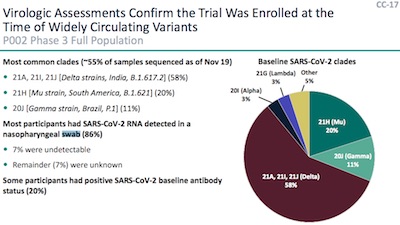
- They also checked that the enrolled subjects had a wide variety of circulating variants of the SARS-CoV2 virus. As shown in the pie chart, the majority was the annoyingly infectious Delta strain, but 5 or so other strains put in noticeable appearances. So any result is likely to be a broad result against SARS-CoV2, not just some particular strain.
- There were 10 subject deaths during the trial, but 9 were in the placebo arm. As a
crude indication of safety, that’s pretty good. There were a lot of other safety
studies, both in vitro, in animals, and in surveillance of the trial population.
The trial seemed pretty safe, with adverse events in the placebo arm about the same as
treatment. There is some worry about mutagenicity, particularly during organogenesis
in pregnancy when messing with human RNA could be a bad idea.
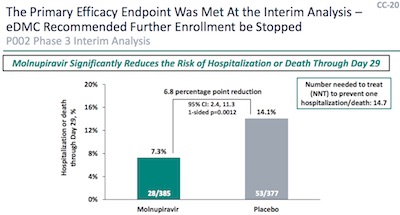
- The initial stopping of the trial was based on an intermediate sample, and showed efficacy vs hospitalization of 48.3% (CL: 20.5% - 66.5%).
-
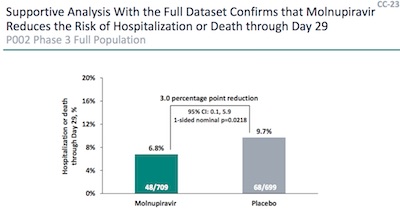
However, the entire dataset gives a more muted effect. From the data shown here for the full cohort, we can reproduce our earlier calculation:
> round(efficacyAndCL(709, 48, 699, 68), digits = 3) LCL Eff UCL 0.010 0.304 0.511So that means molnupiravir has an efficacy against hospitalization of 30.4% (CL: 1.0% - 51.1%). That is both a dramatically lower median efficacy, as well as a wider confidence limit. Apparently the back half of the clinical trial didn’t so so well?! It’s somewhat disappointing, especially in comparison with the robust results of paxlovid (though that’s also an intermediate analysis, I believe). Still, it’s statistically significant (the CL is – barely – bounded above 0%, and a median 30% reduction in hospitalization is nothing to sneeze at!).
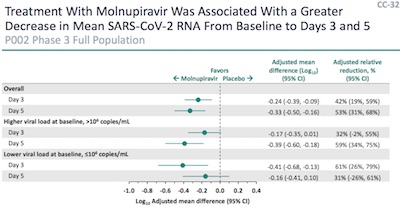
- There is no doubt this improvement is due to virus clearance, since they measured SARS-CoV2 RNA at baseline, day 3, and day 5. The reduction, as measured by log of mean difference and its 95% CL, is significant as shown here. It’s not just making people feel better, it’s doing so by a mechanism that makes sense and is related to the disease process.
A quick flip through the Merck briefing document reveals a lot more information, mostly details. But 2 things were striking to your humble Weekend Editor, surprised that they were left out of the slides:
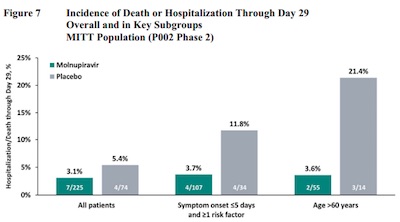
- This graph shows the risk reduction in all patients, patients with some comorbidities, and the elder patients. Note that the gray bars (risk in control group) go up as you move across, i.e., some groups have higher risks. The green bars (risk in the treatment group) are all lower, and comparable. Basically molnupiravir reduced the risk in all of those groups down to a baseline constant. Any difference in counts among the group was not because molnupiravir didn’t work, but because it was facing a higher baseline risk. That’s good to know, that there aren’t resistant subgroups.
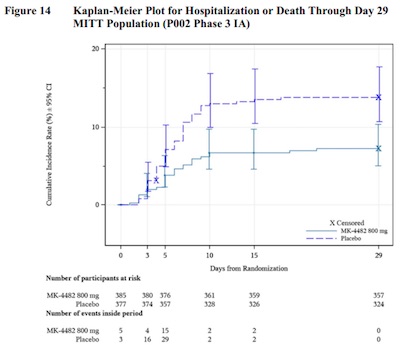
- This is the de rigeur Kaplan-Meier plot. It shows hospitalizations versus time, for the treatment arm and control arm. There’s a log-rank statistic that shows this difference is significant, i.e., the spread between the 2 curves is real.
That’s… kind of convincing to me. (Though keep in mind I skipped most of the toxicity and mutagenicity arguments, so that might change my mind if I had the energy to pay attention to it. Aaaand… that turns out to be the case, sadly enough. See below.)
The FDA presentation
 Looking through the FDA slides, I’m struck by 3 aspects:
Looking through the FDA slides, I’m struck by 3 aspects:
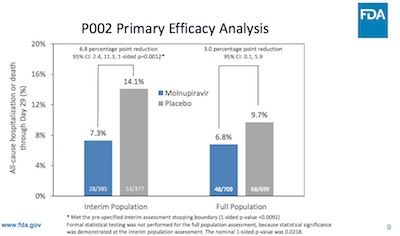
- They came to largely the same conclusions about efficacy vs hospitalization. For example, here are the hospitalization rates in treatment and control arms, for the interim and full cohorts. They come to the same conclusion as Merck, down to the quantitative level. This is what you want to see.
- The FDA spent a lot more time analyzing the safety data, as far as toxicity, mutagenicity, and reproductive effects. If you want to read about the tox stuff that your humble Weekend Editor was too tired to wade through, this is where to jump in.
- The FDA spent a lot more time noodling over the exact patient population for whom molnupiravir should be authorized, in terms of both likelihood of benefit and type of risk (bone marrow, mutagenicity, reproductive abnormalities, requiring pregnancy tests…).
(See below, in the voting: it was a mistake on my part to get bored and go grocery shopping during the tox portion. It turns out there were major reservations about mutagenesis during pregnancy!)
Discussion and voting
Honestly, I skipped over the public comment and committee discussion periods. After almost 2 years, I’m getting COVID-19 fatigue.
The voting took place around 4:45pm EST, which is cutting it fine since the meeting was supposed to end at 5pm. This is how these things always go, though: too much material for the time, and too many opinionated people who all want to talk. But their opinions are highly trained, so it sometimes behooves us to listen. Someday, we’ll learn to allocate the right amount of time to do so.
They used a Zoom breakout room for voting, though I’m somewhat confused as to why. Perhaps to show their votes to each other, but not the public? Or perhaps they’re just tired of being in the public eye all day?
 The result: 13 Yes, 10 No, and 0 Abstain. Here’s a fuzzy screen capture from the YouTube
broadcast, where you can just about make out the names of who voted which way (click to
embiggen). If you watch the video broadcast near the end, each member had to state their
name and their vote and possibly some reasons. So if you want to know who went which way,
that’s where to look.
The result: 13 Yes, 10 No, and 0 Abstain. Here’s a fuzzy screen capture from the YouTube
broadcast, where you can just about make out the names of who voted which way (click to
embiggen). If you watch the video broadcast near the end, each member had to state their
name and their vote and possibly some reasons. So if you want to know who went which way,
that’s where to look.
- The cautionary notes seemed to be around pregnant women, where some mutagenic effects particularly during organogenesis in the fetus were likely (so not in the 1st trimester). If you’re a woman, a prescription for molnupiravir will be preceded by a pregnancy test.
- They also felt it should be limited to high-risk individuals (possibly mostly the unvaccinated or those who had a suboptimal response to vaccination due to immune impairment), because of some other tox effects (which I now regret skipping over!).
- There was also some concern that the induction of high mutation rates in the virus might, if it doesn’t go far enough, create a troublesome new variants.
- The fact that the early part of the clinical trial went so well, while the later part seemed to damp down that result, made people wonder if there was a particular population of responders who should be characterized. That, at least, would mean only those who might benefit from it need take the risk.
- The logistical difficulties of getting the drug to people within 5 days of symptoms looked daunting to some AMDAC members.
Interestingly, given the looming hearings for paxlovid: if another orally available agent with a better risk/benefit profile were to become available, then AMDAC thought the FDA could reconsider approval. So maybe molnupiravir gets approved for a couple weeks until paxlovid? That would be unusually dramatic; a better course would have been to hold the paxlovid hearings first.
Weirdly, absolutely nobody said the word “paxlovid”. Maybe a legal reason for that?
At the end, the chair said: “In my many years of chairing these meetings, this is the first one that’s gone over time. I think that speaks to the complexity of the decision.”
The Weekend Conclusion
Well, it looks like molnupiravir is recommended for EUA, though with less than complete enthusiasm. That’s fair, I guess: the benefit was less than the stellar interim analysis, and the mutagenic effects during pregnancy give one pause. The potential mutagenic effects in pregnancy really rang the AMDAC like a bell.
Now let’s see (a) what the FDA administration does, and (b) whether anybody accelerates the paxlovid hearing!
Multae apologiae for my rash decision to go grocery shopping instead of listening to the tox stuff. Maybe later when I’m not so tired I’ll go back and wade through the tox details.
Addendum 2021-Nov-30 evening:
 Now our safari guides at STAT News check in! [8] Matthew
Herper reports, as above, that AMDAC members were concerned about birth defects and that
the efficacy was not as stellar as initially thought.
Now our safari guides at STAT News check in! [8] Matthew
Herper reports, as above, that AMDAC members were concerned about birth defects and that
the efficacy was not as stellar as initially thought.
In the end, panelists narrowly voted that the benefits of having an oral Covid treatment to keep people out of the hospital outweighed their questions and concerns. But the FDA may write a far narrower authorization for the drug than observers would previously have expected.
Sigh.
Addendum 2021-Dec-02: The efficacy in the back half of the trial
The efficacy of the first cohort was so good the trial was stopped because that was enough for approval and it would be unethical to continue giving placebos instead of just moving ASAP to approval.
But… the final report had only modest efficacy. What does that say about the back half of the trial?
Somebody asked me about that and I looked into it (after first doing it wrong, which is why the linked post is labelled “MeaCulpa”.
It turns out from the slides above you can work all this out. The median efficacies and their 95% confidence limits in the full, interim, and completion cohorts were:
| Cohort | 95% LCL | Efficacy | 95% UCL | |||
|---|---|---|---|---|---|---|
| Full | 1.0% | 30.4% | 51.1% | |||
| Interim | 20.4% | 48.3% | 66.5% | |||
| Completion | -151.6% | -32.5% | 30.1% |
So yeah, the second half of the trial population (the “completion” cohort) was pretty miserable. There were more hospitalizations in the treatment arm than in the control arm (but only 20 vs 15), so the efficacy was negative! Now you see why the AMDAC members were so concerned.
Notes & References
1: FDA Staff, “November 30, 2021: Antimicrobial Drugs Advisory Committee Meeting Announcement”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. The meeting materials[↩] are further down the page.(#fn1a)
2: FDA Staff, “Antimicrobial Drugs Advisory Committee (AMDAC) Meeting: November 30, 2021 Agenda”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. ↩
3: FDA Staff, “Antimicrobial Drugs Advisory Committee (AMDAC) Meeting: November 30, 2021 QUESTIONS”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. ↩
4: S Curtis, D Hazuda, K Blanchard, N Karsonis, “Molnupiravir: U.S. Food & Drug Administration Antimicrobial Drugs Advisory Committee November 30, 2021”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. ↩
5: Merck Staff, “Center for Drug Evaluation and Research, Antimicrobial Drugs Advisory Committee Meeting Briefing Document: Molnupiravir, Oral Treatment of COVID-19, APPLICATION NUMBER: EUA #000108”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. There is also a 7-page addendum. ↩
6: J Farley, A Hodowanec, M Seaton, R Heflich, P Harrington, E Donaldson, J O’Rear, D Birnkrant, “FDA Presentation for Antimicrobial Drugs Advisory Committee Meeting Briefing Document: Molnupiravir, Oral Treatment of COVID-19”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. ↩
7: FDA Staff, “U.S. Food and Drug Administration, Center for Drug Evaluation and Research, FDA Briefing Document: Antimicrobial Drugs Advisory Committee Meeting November 30, 2021”, FDA AMDAC 2021-Nov-30 Materials, retrieved 2021-Nov-30. There is also a 3-page addendum and a 1-page erratum. ↩
8: M Herper, “FDA panel narrowly recommends authorization of first antiviral pill to treat Covid-19”, STAT News, 2021-Nov-30. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.