COVID-19: Is it 'over'?
Tagged:COVID
/
MathInTheNews
/
PharmaAndBiotech
/
Statistics
With vaccination rates rising (albeit glacially slowly), and new therapeutics like molnupiravir and paxlovid about to be approved, people are asking: is it over? My take is: probably not.
Is it over?
We’ve offered some praise in the past for COVID-19 therapeutics that really seem to work. The leaders right now for early intervention seem to be fluvoxamine, molnupiravir and paxlovid. Fluvoxamine is already approved (as an anti-depressant and OCD therapy), while molnupiravir and paxlovid will come before the FDA after a week or two more of review. They are likely to be approved by December, unless something horrific is uncovered, like fraud – and the chances of that are low. (The FDA VRBPAC meets 2021-Nov-30 to consider molnupiravir; paxlovid is not yet scheduled.)
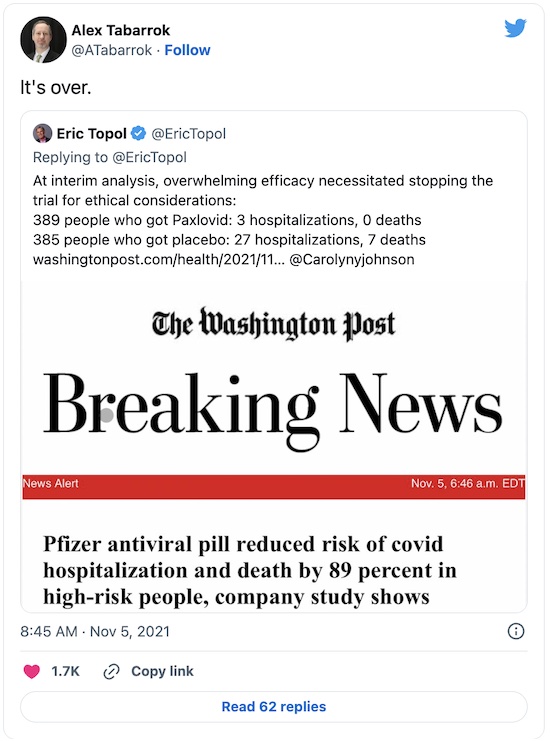
The are, indeed, exciting news! Paxlovid in particular, with an efficacy against hospitalization of 89% (CL: 66% - 96%) looks like it can really move the needle. Molnupiravir, with efficacy of 48% (CL: 21% - 66%) is lower, but still worthwhile. Even more exciting is that all 3 drugs (especially fluvoxamine, which has been widely prescribed for years) seem quite safe.
That means it will be almost inevitable to try them in combination, to look for synergy of effect: getting more than the sum of the individual effects of each drug. True, the companies making them are different, and hence uninterested in “promoting the competitors”, as my erstwhile management would say to my great frustration (I was the guy hunting down synergy combinations in oncology research).
Still, someone will do it. I wouldn’t be even slightly surprised to hear that the NIH was getting grant proposals on this already.
That’s a whacking big bolus of good news. Sure, we have to get molnupiravir and paxlovid approved, but people are on that right now. Sure, we have to do the combination clinical trials. Sure, we have to preposition fluvoxamine, molnupiravir and paxlovid at every pharmacy in the US and then every pharmacy in the world. Sure, we have to educate clinicians and even pharmacists to dispense them.
But we know how to do that. (And we’ve recently proven with the vaccine rollouts that we know how to screw it up.) But, after that… is it over?
Prominent people think so:
But then, prominent people always think so. That is especially true when they’re estimating the difficulty of problems not in their own fields. (Tabarrok is justly famous as an economist, not as a public health specialist.)
Your humble Weekend Editor is not a public health specialist either, but at least somewhat adjacent in pharma research for many years. I think it’s not over.
But why not?!

 A couple WaPo articles by Carolyn Y
Johnson [1] [2]
outline the state of the world and some of the problems ahead.
A couple WaPo articles by Carolyn Y
Johnson [1] [2]
outline the state of the world and some of the problems ahead.
Yes, molnupiravir and paxlovid work. Yes, the safety profiles look pretty good so far. Yes, that means we might be able to use them prophylactically on the contacts of an infected person before they show symptoms. Yes, Pfizer has already announced they will allow immediate generic manufacture of paxlovid abroad. Yes, the US government has pre-ordered millions of courses of treatment even before approval, to make sure manufacturing gets spun up quickly. Yes, manufacturing is in fact spinning up:
- US pre-purchases: 3.1 million courses from Merck and 10 million from Pfizer
- Pfizer manufacturing plans: 50 million courses in 2022
- Merck manufacturing plans: 10 million by end of 2021, “more” in 2022
But… these aren’t a complete solution to the problem. A piece, yes. But other pieces like rapid testing, vaccination, boosters, antibodies, increased hospital ventilator capacity, and so on cannot be neglected. The main role of these 2 new antivirals is if there’s lots of rapid testing, then you get your infection caught, diagnosed, and prescribed early; then you spend a week at home taking a couple pills a day so your infection is mild. During that time you still have to isolate, with which lots of people will refuse to comply.
 A slightly more realistic (and pessimistic) take on the situation comes from Lev Facher at
STAT News. [3] He points out a number of structural
problems in the US healthcare system that in effect require people to jump through
multiple hoops to get treatment: enough health education to recognize symptoms, finding a
test, getting tested and diagnosed, getting a prescription from a doctor, and getting that
prescription filled. This introduces multiple points of failure, and is made worse by
economic inequality, structural racism, and language barriers. We make it hard for some
groups to access health care.
A slightly more realistic (and pessimistic) take on the situation comes from Lev Facher at
STAT News. [3] He points out a number of structural
problems in the US healthcare system that in effect require people to jump through
multiple hoops to get treatment: enough health education to recognize symptoms, finding a
test, getting tested and diagnosed, getting a prescription from a doctor, and getting that
prescription filled. This introduces multiple points of failure, and is made worse by
economic inequality, structural racism, and language barriers. We make it hard for some
groups to access health care.
Just consider testing. Tests are in scandalously short supply in the US:
- They’re not free, like in the UK. If you’re poor, the cost is significant.
- You have to go to multiple stores to find one that has them in stock, which can take hours; alternatively, you can wait in line at a clinic which may only be open during your work hours or which may also require you to wait for hours with other possibly-sick people.
- The test has to be evaluated; for PCR tests this involves at least a 24 hour delay, and sometimes several days (at which point the information is more or less useless).
- You have to get an appointment with your PCP which may be weeks in advance; if you’re poor or minority, you may not even have a regular PCP and insurance.
- You have to purchase the drugs, which without insurance will be a great burden for the poor.
And all of that has to happen within about 3 – 5 days of the onset of symptoms. While you’re feeling sick, you must first fight the sclerotic US healthcare system.
Facher points out, I hope ironically, that the best places for that to happen are nursing homes and prisons: the person doing the testing, diagnosis, prescribing, and administration may well be the same person on the same day.
Conclusion: In order to make early treatment work, testing must be fast, cheap, and ubiquitous.
I can think of a number of other stumbling blocks that we’ve managed to stumble over in the past, so they’re possible this time:
- For paxlovid, there are still pending several clinical trials testing for common drug interactions. The trial excluded people with a history of liver/kidney impairment, or drugs that mess with P450. We might find a problem there, or the FDA might delay until those trials are done.
- The disinformation machine has already swung into motion, spewing forth the usual intellectual sewage. “Molnupiravir will alter your DNA.” Or, “it’s just ivermectin”, repackaged at a higher price (which Reuters debunked twice). (Can we get a new term for purveyors of disinformation, like “info-terrorists”, or something?)
-
Some ninnies have already decided that since there are treatments available, they no longer need even consider getting vaccinated. In the words of an anonymous doctor quoted in the WaPo articles:
Syphilis is treatable with penicillin. But it is far better to not get it in the first place.
Do we really have to explain to people that getting COVID-19 through inattention to vaccination is about as dumb as getting syphyilis through inattention to safe sex? Apparently so.
- The people unable to access vaccinations (or who refuse it), and are unable to access the new antivirals might be a breeding ground for a new variant worse than Delta.
Yeah, I’m in a more pessimistic mood (true, it’s a character flaw). But I wish I didn’t keep getting confronted with evidence that pessimism is merited by the situation.
The Weekend Conclusion
We’ll get to herd immunity when > 85% are vaccinated or recovered from COVID-19, and the rest are dead. (The 85% comes from applying a SIR model to the Delta $R_0 \sim 6 - 7$ or so.) Because those are the inevitable near-term alternatives: either you get vaccinated, or you will get Delta COVID-19, after which you are (somewhat the worse for wear) recovered… or you are dead.
Until herd immunity, here at Chez Weekend we prefer to sort out the probabilities to bias our future against the “being dead” part. That means getting vaccinated to prevent COVID-19 and make any breakthrough infection likely milder, as well as checking health insurance is up to date and that we’re on good terms with our PCP.
You should consider doing likewise.
Notes & References
1: CY Johnson, “Antiviral pills from Pfizer, Merck, show promise against worst covid-19 outcomes”, Washington Post, 2021-Nov-05. ↩
2: CY Johnson, “Treatments will change the pandemic, but they can’t end it alone”, Washington Post, 2021-Nov-21. ↩
3: L Facher, “Covid antivirals could be pandemic game-changers. But Americans might struggle to access them”, STAT News, 2021-Nov-23. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.