The AstraZeneca/Oxford Vaccine vs Blood Clots
Tagged:COVID
/
JournalClub
/
MathInTheNews
/
PharmaAndBiotech
/
R
/
SomebodyAskedMe
/
Statistics
Somebody asked me whether worries the AstraZeneca/Oxford vaccine and dangerous blood clotting seen in some patients were a real problem. You know what the anti-vaxxers are gonna say; what do the data say?
What’s the sitch?
The Bayesian prior on AZ/OX here at Chez Weekend
Here at Chez Weekend, we haven’t been fans of the AstraZeneca/Oxford vaccine for COVID-19. The Phase 3 trial that read out last December had numerous problems:
- A CMO bungled the dosing, delivering half doses instead of full doses.
- AZ didn’t assay the CMO product to check, and didn’t randomize doses from different batches across test sites. Efficacy was hopelessly convolved with batch.
- So they got radically different results in the South African variant sites, but averaged them with other sites anyway in spite of the glaring differences. If there’s no near superclass of all the things being averaged, you can’t do it: it’s like averaging apples, oranges, … and sledgehammers.
- They failed to disclose any of this; the CEO even attempted to spin it as “not a mistake” because they amended the trial protocol. That’s like taking a pratfall and saying “I meant to do that!”
AZ/OX is now in another clinical trial [1], this one hopefully with more transparency and a clean readout. It’s probably pretty good, but we need evidence before the FDA will look at it. In fact, word on the street — assuming you travel the appropriate streets — is that this trial has reached its required event count and will read out in the next few days:
So when I heard that some European countries were stopping the use of AZ/OX pending an assessment of serious adverse events (occasionally fatal blood clots) I thought, “What have they done wrong now?” (‘They’ meaning either AstraZeneca or the governmental authorities.) Either the vaccine is dangerous and lives are saved by not using it, or the vaccine is ok and lives are saved by using it. Which is it?
Some terminology
They’re complaining about thromboembolic events, so we’d best start by getting all of us reminded and on the same page about the meaning of thromboembolism. Basically, some blood partially or fully clots inside the circulatory system (a thrombus), which blocks something else (an embolism):
- VTE: veinous thromboembolism, if it forms in a vein.
- DVT: deep vein thrombosis, a subtype of VTE when it forms in a limb, usually a leg. A common worry for older people who take long flights in coach class.
- PE: pulmonary thrombolism, when a DVT embolism is “thrown” and blocks finer vessels in the lung (very serious, as in sudden death).
- CSVT: cerebral sinus vein thrombosis, a subtype of VTE, like PE, but when a clot is thrown into the brain (somewhat rare, very bad news when it happens).
- ATE: somewhat rarer case where a thrombus forms in an artery (doesn’t seem to be the case here).
So we’re worrying about some observations of VTE (either DVT or PE) in some recent recipients of the AZ/OX vaccine.
Background as seen in the general media

 For some background in general media, high-brow and mid-brow, we turn to articles in the
New York Times [2] and
BuzzFeed [3]. The content is pretty similar, in spite of
differences in tone:
For some background in general media, high-brow and mid-brow, we turn to articles in the
New York Times [2] and
BuzzFeed [3]. The content is pretty similar, in spite of
differences in tone:
- There does not appear to be any relationship to the vaccine at all.
- According to Prof Stephan Moll, of UNC medical: in the US each year there are 300,000 - 600,000 cases of VTE (DVT or PE), so that’s very roughly 100-200 cases/day. The US is vaccinating about 1% of the population every day, so we should expect to see about 10 - 20 VTE cases/day in newly vaccinated people, just because that’s one of the things that happens to people, vaccine or no. It’s not that rare!
- Apparently one of the European sites saw a CSVT. Those are rare enough to make people take notice, possibly explaining some of the worry here.
Field findings
What the regulatory agencies and AstraZeneca have to say


 Ok, that sort of tells us the kind of thing we’re looking at, but what about the
specifics in the AZ/OX vaccination population? Let’s avoid the media entirely here, and
look at primary sources: the European Medicines
Agency (EMA) [4] [5] and
AstraZeneca itself [6]:
Ok, that sort of tells us the kind of thing we’re looking at, but what about the
specifics in the AZ/OX vaccination population? Let’s avoid the media entirely here, and
look at primary sources: the European Medicines
Agency (EMA) [4] [5] and
AstraZeneca itself [6]:
- The EMA says it’s investigating whether there are enough thromboembolic events to worry about the vaccine, but there is so far no indication the vaccine is at fault. The stoppages are out of “caution”, though it seems to me that “caution” should include the number of deaths due to people going without COVID-19 vaccination for a few more days/weeks, too!
- Specifically, they say there were 30 throboembolic events out of 5mln people vaccinated as of 2021-Mar-10.
- The first thing anybody would check would be if the events were all at a small number of sites. If so, and if those sites all received the same batch of vaccine, you’d suspect the batch itself. So far, no evidence for a batch-specific problem.
- AstraZeneca fills in a couple details: 15 DVT + 22 PE = 37 VTE events as of 2021-Mar-08, out of ~ 17mln vaccinees in EU+UK as of 2021-Mar-08. No idea why they say 37 when the EMA says 30, for reporting 2 days later. But let’s pessimistically go with the higher number of 37 VTE events.
- In the clinical trial, VTE events were lower in treatment arm, because COVID causes blood clots & control arm caught more COVID. So in that sense you can regard the vaccine as protective against VTE events.
- No batch effect observed, after extensive QC by AstraZeneca of the batches shipped to the sites where VTEs were observed.
Summary: So far, nobody has actual evidence to connect the vaccine to the VTE events. The stoppages are out of (possibly misplaced?) caution. The numbers seem to be 30-37 VTE events out of ~ 5 mln vaccinated. That means the probability of VTE is $\Pr(\mathrm{VTE} | \mathrm{vax}) = \frac{37}{5,000,000} = 7.4 \times 10^{-6}$ … or about 7 times in a million patients. Reasonably rare, and seems like a risk worth taking compared to COVID-19, even if we were guaranteed that many VTEs!
Does COVID itself cause thromboembolic events?


 One question we want to consider is whether COVID-19 causes thromboembolisms. If so, then
even if the vaccine did increase the risk slightly, the fact that COVID-19 increases the
risk a lot is a good reason to take the vaccine. Not taking the vaccine doesn’t mean
your VTE risk is like normal times; it means your risk is like pandemic times, where the
alternative is all the risks of catching COVID-19.
One question we want to consider is whether COVID-19 causes thromboembolisms. If so, then
even if the vaccine did increase the risk slightly, the fact that COVID-19 increases the
risk a lot is a good reason to take the vaccine. Not taking the vaccine doesn’t mean
your VTE risk is like normal times; it means your risk is like pandemic times, where the
alternative is all the risks of catching COVID-19.
So does COVID-19 cause VTEs? Apparently yes! [7] [8] [9]
- In a New Orleans cohort, VTE events tended around 3.1% in all hospitalized COVID-19 patients, and around 7.2% for those on ventilation. Those are incredibly high numbers, compared to the background population, where on any given day 3% - 7% of the population does not have VTE!
- In a meta-analysis of 36 other studies, it was found that the chance of VTE and the 95% confidence limits were: 10% (CL: 6% - 14%) outside the ICU, and 28% (CL: 22% - 34%) for those in the ICU. Again, incredibly high!
- The cause appears to be the cytokine storm from the immune response to SARS-CoV-2. This can also cause thrombocytopenia (low platelet count), which leads to clots.
So… yeah, COVID-19 causes lots of inconvenient and potentially lethal clots. Avoiding that risk, you should look very charitably and forgivingly at a vaccine’s alleged clot risk. (Even if there were some evidence of vaccine-induced thromboembolism, which we still haven’t established! )
How often does thromboembolism happen in the general population?

 Of course, to do a proper analysis here, we want to know how often VTE occurs in the
general population (unvaccinated, pre-COVID days). We want our vaccine to have a risk of
VTE no higher than that background rate, if we possibly can. We wanted both a
practice-oriented view and a science-oriented view, so we consulted papers from the
American Journal of Managed Care [10]
and Nature Reviews Cardiology [11].
Of course, to do a proper analysis here, we want to know how often VTE occurs in the
general population (unvaccinated, pre-COVID days). We want our vaccine to have a risk of
VTE no higher than that background rate, if we possibly can. We wanted both a
practice-oriented view and a science-oriented view, so we consulted papers from the
American Journal of Managed Care [10]
and Nature Reviews Cardiology [11].
- The AJMC practice-oriented paper quoted VTE frequencies in the general population of 1-2 per 1,000 person-years. It also noted, chillingly enough, that the “presenting manifestation of PE is sudden death”. Sounds skippable, if skipping is an option…
- Nat Rev Cardiol agreed: VTE frequency was 104 – 183 per 100,000 person years, which on the same scale as the preceding paper is 1.04 – 1.83 per 1,000 person-years — so pretty consistent. Notably, it is higher in African Americans, but lower in Asians, Asian Americans, and Native Americans.
Quite relevantly, VTE frequency is higher in the elderly, who are the population getting vaccinated as early as possible now. So using 1-2 per 1,000 person-years is an underestimate of the VTE frequency in the current vaccine population. But since that imposes an even more stringent threshold to beat, let’s go with it.
Some statistical hypothesis tests
We now have enough data to compute the probability per unit time (the rate) at which VTE events occur in both the general population and in the recently vaccinated population.
- Are they statistically significantly different?
- If so, favoring which group with a lower rate?
- By how much?
When we can answer those questions, quantitatively, we’ll be able to form an opinion about whether AZ/OX is related to clotting disorders. It’ll be a preliminary opinion, since the safety review boards will have full access to patient medical records for a detailed, deep dive which we cannot do with overview numbers. But we can tell which way to bet!
Let’s assume the observation time after a vaccination is 2 weeks. Then our rates of VTE/fortnight are: \(\begin{align*} d/dt \Pr(\mathrm{VTE} | \mathrm{bkgnd}) & = (1 \mathrm{VTE} / 1000 \mathrm{person-years}) \times (1 \mathrm{year} / 26 \mathrm{fortnights}) \\ & = 1 \mathrm{VTE} / 26,000 \mathrm{person-fortnights} \\ & = 3.8 \times 10^{-5} \mathrm{VTE/person-fortnight} \\ d/dt \Pr(\mathrm{VTE} | \mathrm{vax}) & = 37 \mathrm{VTE} / 5,000,000 \mathrm{person-fortnights} \\ & = 7.4 \times 10^{-6} \mathrm{VTE/person-fortnight} \end{align*}\)
NB: the rate of VTEs is lower in the vaccinated population, by a factor of 5. In other words, the top-line number says the vaccine is protective against VTE.
We know there were about 5,000,000 AZ/OX vaccination subjects. What should we use as the corresponding size of the background population? Since we’re looking at a vaccination campaign in Europe, the population of the European Union (446 million) looks like about the right thing.
Now we can build a crosstabulation showing the VTE rates in the vaccinated population and the unvaccinated population. We use $\Pr(\mathrm{VTE} | \mathrm{background})$ and the EU population above to compute the background counts to show a rate of about 1/26,000 every 2 weeks in the background population:
> popln <- 446000000
> mx <- round(matrix(c(37, 5000000, popln * 1/26000, popln), byrow = TRUE, nrow = 2, dimnames = list(c("Vax", "Bkgnd"), c("VTE", "No VTE")))); mx
VTE No VTE
Vax 37 5.00e+06
Bkgnd 17154 4.46e+08
We next apply Fisher’s exact test (devised, according to legend, for the problem of The Lady Tasting Tea) and the somewhat more traditional test of proportion to test the statistical significance of the difference in rates of VTE between the vaccinees and the general EU population. For both tests, the null hypothesis is that there’s no difference:
> fisher.test(mx)
Fisher's Exact Test for Count Data
data: mx
p-value < 2.2e-16
alternative hypothesis: true odds ratio is not equal to 1
95 percent confidence interval:
0.1354152 0.2653069
sample estimates:
odds ratio
0.192402
> prop.test(mx)
2-sample test for equality of proportions with continuity correction
data: mx
X-squared = 124.34, df = 1, p-value < 2.2e-16
alternative hypothesis: two.sided
95 percent confidence interval:
-3.361408e-05 -2.850614e-05
sample estimates:
prop 1 prop 2
7.399945e-06 3.846006e-05
Both tests report ridiculously strong significance, at $p \le 2.2 \times 10^{-16}$, the lowest $p$-value that R will report without embarrassment. That says yes, we really should believe the VTE risk is lower among the vaccinees.
Finally, we can add our usual Bayesian analysis. We’ve experimentally measured $\Pr(\mathrm{VTE} | \mathrm{Vax})$ and $\Pr(\mathrm{VTE} | \mathrm{bkgnd})$. Each of those should be binomially distributed, with some underlying success parameters $p_{\mathrm{Vax}}$ and $p_{\mathrm{bkgnd}}$. The usual Bayesian analysis says that if we start with a uniform prior on each $p$, then the posterior is a Beta distribution. We can calculate & plot those distributions, to see if they look different:
> source("../../tools/graphics-tools.r")
> ps <- seq(from = 0, to = 5.0e-5, length.out = 1000)
> vax <- dbeta(ps, shape1 = 38, shape2 = 5.00e+6)
> bkgnd <- dbeta(ps, shape1 = 17155, shape2 = 4.46e+8)
> withPNG("../images/2021-03-17-azox-vaccine-thrombo-beta.png", 600, 300, FALSE, function() { withPars(function() { matplot(ps, matrix(c(vax, bkgnd), byrow = FALSE, ncol = 2), type = "l", lty = "solid", col = c("blue", "black"), xlab = "p", ylab = "Density", main = "Bayesian Posterior Beta Distributions"); legend("topright", inset = 0.01, bg = "antiquewhite", legend = c("Vax", "Bkgnd"), col = c("blue", "black"), lty = "solid", lwd = 2) }, pty = "m", bg = "transparent", ps = 16, mar = c(3, 3, 2, 1), mgp = c(1.7, 0.5, 0)) })
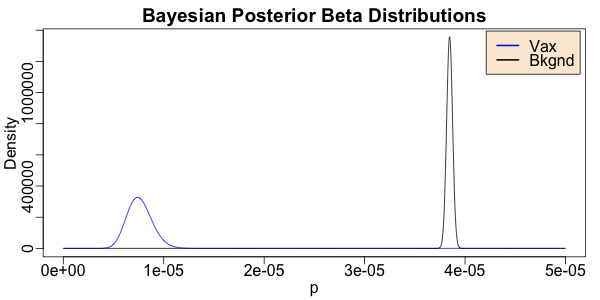
These 2 resulting Beta distributions tell us what we should believe about the probability of VTE in the general population (black) and among the vaccinees (blue). They tell us not just the probability at the peak, but also the uncertainty around each. It’s crucial when reporting a result to be clear about uncertainty! The bottom line is that:
- The 2 distributions are well separated. This confirms the statistical significance result above, namely that the VTE frequency is different among vaccinees and the general population.
- The rate of VTE among vaccinees is lower than in the general population. So yes, there is an effect; but it’s protective against VTE in the vaccinees.
Conclusion: Based purely on the top-level numbers – the counts of VTE and non-VTE vaccinees and the rate of VTE in Europeans – it looks like these events are not related to the vaccination, and indeed that the vaccination may be protective against VTE.
Some safety judgements from experts
Ok, so that’s what one retired scientific loudmouth with a blog thinks. There’s no particular reason you should care. What do real experts think?
- Reuters reports [12] that the World Health Organization generally appealed to country authorities not to stop using the AZ/OX vaccine. Given that it’s been dosed in 5 million people and the side-effects are in line with the general population, it’s provably safe and the observed adverse effects show no relation to the vaccine. Given the extreme risks of COVID-19, it’s much better to take the vaccine than not.
- New Scientist reports [13] something similar, saying the decisions of governments to suspend use are not rational. In particular, they note that the rate of VTE is not higher than the general population, and COVID-19 itself poses significant thromboembolism risks:
“Many thousands of people develop blood clots annually in the EU for different reasons,” the EMA said in a statement. The number of blood clotting incidents in vaccinated people “seems not to be higher than that seen in the general population”.
“There is absolutely no data that supports [the German government’s] decision,” says César Muñoz-Fontela at the Bernhard Nocht Institute for Tropical Medicine in Germany. He says that older people and people with pre-existing health conditions, who are more at risk of blood clots generally, have been prioritised for the vaccine, which may have skewed the apparent side effects. He would like to see a comparison with a control group that has the same characteristics as the people so far vaccinated.
The International Society on Thrombosis and Haemostasis recommends that all eligible adults continue to receive their covid-19 vaccinations. “At this time, the small number of reported thrombotic events relative to the millions of administered COVID-19 vaccinations does not suggest a direct link,” it said in a statement.
“In weighing up the merits of a medical intervention, it’s really important to consider both sides of the argument: how risky is it for someone to have it versus how risky is it for them not to,” says Lucy Walker at University College London. “An increased risk of thrombosis is one of the known complications of [coronavirus] infection. The vaccines we have are incredibly good at preventing the illness caused by this virus. They will therefore prevent people from having thrombosis associated with the infection itself.”
- The EMA safety committee investigates [14], finds no effect so far (emphasis in original):
While its investigation is ongoing, EMA currently remains of the view that the benefits of the AstraZeneca vaccine in preventing COVID-19, with its associated risk of hospitalisation and death, outweigh the risks of side effects.
So it looks like “no link of VTE events to AZ/OX vaccine” is the consensus opinion of experts, not just loudmouth retired scientist bloggers. Maybe you should believe that.
The Weekend Conclusion
More and more people die from COVID every day; that’s the risk you need to beat.
Thinking specifically about thromboembolisms, we’ve seen here that the AZ/OX vaccine appears to lower the risk compared to the general population. And since it prevents most COVID-19 which would skyrocket the rate of VTE, it seems slam-dunk-obvious that the vaccine is A Good Thing to Do. Withholding the vaccine means more people are at risk of COVID-19, and more deaths.
There is further risk that people will become superstitiously fearful of the AZ/OX vaccine, and reject it entirely. This is not a theorietical risk; there’s actual precedent for the Lyme disease vaccine from GSK. People associated it with arthritis even though the numbers said it was just random. Enough people refused to take it, that the vaccine was removed from the market, leaving the population exposed to Lyme disease. Grady & Robbins in the NYT piece, op. cit., tell the story:
After the Food and Drug Administration approved a highly effective vaccine for Lyme disease in 1998, media reports and a class-action lawsuit gave fuel to complaints from people who said they had developed arthritis after being vaccinated. In 2001, the F.D.A. reconvened a panel of experts to review the vaccine’s safety data — clinical trials had turned up no difference in the frequency of long-term joint symptoms between the vaccine and placebo groups — and concluded that the vaccine should stay on the market.
But by then, public perception of the vaccine was too far gone: A steep drop-off in sales spurred the vaccine’s maker, GlaxoSmithKline, to pull the vaccine from the market in 2002.
That’s the last outcome we need with a COVID-19 vaccine.
 The formidable med-chem blogger Derek Lowe has also weighed in at
In the Pipeline [15]:
The formidable med-chem blogger Derek Lowe has also weighed in at
In the Pipeline [15]:
Let’s say that the efficacy numbers come in at a solid, inarguable 60%. You would want to see a higher number in a better world, but 60% is a damn sight better than not getting vaccinated at all. Which is effectively what a number of European countries have chosen to do instead. If I were living in one of those countries where the cases are heading right back up, I would bare my arm immediately for a 60% effective vaccine and hope that as many other people as possible did the same.
Indeed.
So why is that not what’s happening? There could be several reasons; I can think of at least 3:
- Maybe they know something we don’t. For all the length of this post, we really only know 4 numbers: the number of observed VTEs in vaccinees (37), the number of vaccinees (5 million), the rate of VTE per fortnight in Europeans generally (3.8e-5), and the population of Europe (446 million). The PRAC knows complete medical histories, and thus may be on the trail of something more subtle that is not visible in the top-line numbers. True, they could be more transparent, but they’re not necessarily doing the wrong thing here.
- Maybe they’re extremely cautious, beyond any ability to compare risks. That is, they see the VTEs in vaccinees and are paralyzed by fear. This is, of course, worthy of our sympathy even though it is not rational.
- Maybe they’re in severe political blame-avoidance mode. One can realize there is no VTE danger posed by the vaccine, but doubt the ability of voters to realize the same thing. While likely true, this position deserves our contempt: politicians who would prefer citizens die of COVID-19 rather than save them with a medically useful but politically perilous vaccine are paralyzed by the basilisk-like gaze of their sociopathic self-regard.
The AZ/OX vaccine, whatever stupidities may have been perpetrated in its name in its first clinical trial, does not look like it’s at fault for clotting adverse events. It should be restored to service as fast as possible, to prevent further COVID-19 deaths.
Addendum 2021-Mar-18: Some details on the thromboses found
 Gary Cornell emailed to ask if we’d seen the news report in
Science on the particular thromboses observed [16], and
why that made the authorities want to suspend use of the AZ/OX vaccine.
Gary Cornell emailed to ask if we’d seen the news report in
Science on the particular thromboses observed [16], and
why that made the authorities want to suspend use of the AZ/OX vaccine.
Well, no… we hadn’t. But that’s one of the things friends & colleagues are for.
Remember above where we said one of the possibilities might be that the regulatory authorities know something we don’t? We didn’t have a particularly high Bayesian prior on that, but we’re updating now to raise the posterior probability on that a bit:
- It seems that although the rate of thromboembolisms are not particularly scary (as we showed here), the types of thromboembolisms are pretty scary. Ok, fair enough: that’s only something one could know with access to detailed medical records, not the top-line numbers that we analyzed here.
- They’re analyzing 13 patients in particular (out of the 37 total VTEs?), in 5 separate countries. That’s a very low level of incidence, so the type had better be really scary.
- They start with immune thrombocytopenia (ITP), which lowers platelet levels and increases clotting. But then it led to cerebral sinus vein thrombosis (CSVT, above), which is a bit unusual — usually it would throw a clot to the lungs (PE), not the brain. This clot blocks the vein that drains the brain, the heart keeps pumping blood in while none drains out, and what you think happens next is what happens next. This is sort of rare as clotting problems go, and to see 7 at one time in a vaccine-related setting is disturbing. That’s what caused the Paul Ehrlich Institute to raise the alarm.
- Also, the CSVT’s developed in younger people: they face acute danger from CSVT, but would have faced less danger with COVID-19 due to their youth. So that’s an imbalance of risk that would be unacceptable if a link were to be proven.
- It’s possible the cases already had COVID-19 when getting vaccinated, as “many” were healthcare workers and teachers. The combined immune reaction could have caused ITP. That, of course, could be fixed with a COVID-19 PCR or antigen test before vaccination.
On the one hand, witholding the vaccine is a problem, as expressed both here and by actual experts:
“The harm caused by depriving people of access to a vaccine will likely vastly outweigh even the worst case scenario if any link to the clotting disorders is eventually found,” University of Leeds virologist Stephen Griffin told the United Kingdom’s Science Media Centre. The European Medicines Agency (EMA) and the World Health Organization have recommended that countries continue immunizations while they investigate the reports.
…
EMA will convene a panel on Thursday to help figure out whether there was a causal relationship between the vaccinations and the highly unusual syndrome. For the moment, the agency is “firmly convinced” that the vaccine’s benefits outweigh any risks, [EMA head Elmer] Cooke said.
On the other hand, the hematologists have now pointed to a very weird increase in CSVT and a link to ITP that may be linked to vaccinating aleady COVID-positive patients. That’s both a specific disease claim and a proposed method of action, so it’s something to take seriously.
Revised conclusion: It still looks to me like over-caution, given the absolute certain risks of COVID-19. But the relevant experts have pointed to something somewhat worrisome. It is probably a fluke, but nobody knows at this point. Maybe stopping the vaccine for a very short time while they investigate looks sensible?
Addendum 2021-Mar-19: AZ/OX vaccinations in Europe resumed
 Various news sources are today reporting that the European authorities have begun using
the AZ/OX vaccine again.
Various news sources are today reporting that the European authorities have begun using
the AZ/OX vaccine again.
To get the most skeptical report, we went to the Paul-Ehrlich-Institut, the German institue for vaccines and biomedicines that first blew the whistle on the unusual number of CSVTs. In today’s press release [17], they quoted the PRAC (Pharmacovigilance Risk Assessment Committee) assessment of the EMA (European Medicines Agency) as seeing a positive risk/benefit trade-off and the addition of a warning of “very rare cases” of CSVT on the label (emphasis added):
The safety assessment by the Pharmacovigilance Risk Assessment Committee (PRAC) at the European Medicines Agency (EMA) confirms the positive benefit-risk ratio of COVID-19 Vaccine AstraZeneca. As a risk-mitigation warning, the SmPC includes the fact that in very rare cases, specific thromboses are found in the period up to 16 days after vaccination. Based on the positive safety assessment by the EMA, Germany will resume vaccination with the COVID-19 Vaccine AstraZeneca starting 19 March 2021. Those willing to be vaccinated will be informed about the above cases during vaccination education. Medical doctors can find information on the website of the Paul-Ehrlich-Institut.
In any large dataset, you’re going to sample the outliers as well as the typical cases: there will always be something outrageous in the low-probability tails. That’s what seems to have been the case here: if you you look carefully by slicing and dicing into all possible subsets of all possible types, eventually something will look statistically significant. Perhaps the trouble could have been avoided by a proper multiple hypothesis test correction (more simply, visually, and brilliantly explained by XKCD) like my favorite, the Benjamini-Höchberg method?
As we saw here, where a delay in vaccination costs lives even when everybody is trying as hard as they can to do good in the world, statistical methods matter.
Addendum 2021-Mar-30: More on a possible clot mechanism
Derek Lowe, at In the Pipeline, has another article on the issue. [18]
People are proposing something called vaccine-induced prothrombotic immune thrombocytopenia (VIPIT). But the latest figures are 31 CSVTs among 2.7 million vaccinees, or a very low probability of 1.15e-5/person. So we’re still talking about rare events, at least, though the normal background according to the Paul Ehrlich Institut would have been 1 or 2 CSVTs. Also, it appears to be happening in younger women (> 50) for whom COVID-19 is less risk. So those 2 facts combined make it of some possible concern, though given the low frequency not enough to stop using the vaccine.
There is no similar signal with Pfizer/BioNTech, Moderna, or J&J vaccines currently widely used in the US. Nor is a similar signal with the AZ/OX vaccine seen in the UK. It seems nobody really knows for sure that something real is happening, but anecdotally it’s concerning.
It might be a rare event worth diverting younger women to another vaccine, or it might all be a case of lack of multiple hypothesis test correction. It’s passed beyond my complexity limit and beyond my competence to have an opinion, in accordance with Scott’s Razor.
Notes & References
1: AstraZeneca, “Phase III Double-blind, Placebo-controlled Study of AZD1222 for the Prevention of COVID-19 in Adults”, Clinical trial NCT04516746, ClinicalTrials.gov, retrieved 2021-Mar-16. ↩
2: D Grady & R Robbins, “Should You Be Concerned About Blood Clots, Bleeding and the AZ-Vaccine?”, New York Times, 2021-Mar-15. ↩
3: P Aldhous, S Lee, “This Is What We Know So Far About AstraZeneca’s COVID-19 Vaccine And Blood Clots”, BuzzFeed News, 2021-Mar-15. ↩
4: European Medicines Agency, “COVID-19 Vaccine AstraZeneca: PRAC investigating cases of thromboembolic events - vaccine’s benefits currently still outweigh risks - Update”, EMA News, 2021-Mar-11. ↩
5: European Medicines Agency, “COVID-19 Vaccine AstraZeneca: PRAC preliminary view suggests no specific issue with batch used in Austria”, EMA News, 2021-Mar-10.↩
6: AstraZeneca, “Update on the safety of COVID-19 Vaccine AstraZeneca”, AstraZeneca Press Release, 2021-Mar-14.↩
7: J Hill, et al., “Frequency of venous thromboembolism in 6513 patients with COVID-19: a retrospective study”, Blood Adv 4:21 (2020), 5373-5377. DOI↩
8: K Boonyawat, et al., “Incidence of thromboembolism in patients with COVID-19: a systematic review and meta-analysis”, Thrombosis Jnl 18:34 (2020). DOI↩
9: S Mondal, et al., “Thromboembolic disease in COVID-19 patients: A brief narrative review”, Jnl Intens Care 8:7 (2020). DOI ↩
10: H Phillipe, “Overview of venous thromboembolism”, Am J Manag Care 23(20 Suppl) 2017-Dec, S376-S382. PMID: 29297660↩
11: J Heit, “Epidemiology of venous thromboembolism”, Nat Rev Cardiol 12:8 (2015-Aug), 464-474. PMC4624298. PMID:26076949. DOI↩
12: S Nebehay, J Gronholt-Pederson, “WHO urges world not to halt vaccinations as AstraZeneca shot divides Europe”, Reuters, 2021-Mar-15. ↩
13: L Liverpool, “No indication AstraZeneca vaccine causes blood clots, says regulator”, New Scientist, 2021-Mar-16. ↩
14: European Medicines Agency, “EMA’s safety committee continues investigation of COVID-19 Vaccine AstraZeneca and thromboembolic events – further update”, EMA News, 2021-Mar-15. ↩
15: D Lowe, “What is Going on With the AstraZeneca/Oxford Vaccine?”, In the Pipeline, 2021-Mar-16.↩
16: G Vogel & K Kupferschmidt, “‘It’s a very special picture.’ Why vaccine safety experts put the brakes on AstraZeneca’s COVID-19 vaccine”, Science, 2021-Mar-17. doi:10.1126/science.abi5259↩
17: Paul-Ehrlich-Institut, “COVID-19 Vaccine AstraZeneca – Safety Assessment Result: The Vaccine is Safe and Effective in the Fight against COVID-19”, Paul-Ehrlich-Institut press releases, 2021-Mar-19. ↩
18: D Lowe, “Blood Clots and the AZ Vaccine, Revisited”, In the Pipeline, 2021-Mar-30. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.