Today: Shot for the 7th time
Tagged:COVID
/
MathInTheNews
/
PharmaAndBiotech
/
Statistics
Today I got my 7th vaccination shot in the last 12 months. Happily.
Nous n’avons pas attrappé le poisson d’avril!
 Honest, this is not an April Fool’s joke. Not even of the delicious French variety (which
always involve fish, for some reason).
Honest, this is not an April Fool’s joke. Not even of the delicious French variety (which
always involve fish, for some reason).
In the last 12 months, I’ve gotten 2 shingles vaccine doses, 1 flu vaccine dose, and now another COVID-19 booster (the 4th COVID-19 vaccination), for a total of 7. That’s a lot of vaccinations. I’ve really been making my immune system pay its rent, but now I can stand in the shadow of its protection.
And that’s A Good Thing.
Here’s the story of the latest COVID-19 vaccine booster, and how I decided to offer up my personal deltoid to be injected (but not inspected or selected, as the anthem of my generation goes).
The Last Couple Days in COVID-19 News
Let’s start from the original sources, then work our way outward into the secondary media with their summaries and analyses, and then some of the scientific literature underlying the decision process.
The Primary Sources

 The primary sources, here in the US, are of course the FDA [1]
and the CDC [2]. They approved a second booster (i.e., 4th
dose) of either the Pfizer/BioNTech or Moderna mRNA vaccines for:
The primary sources, here in the US, are of course the FDA [1]
and the CDC [2]. They approved a second booster (i.e., 4th
dose) of either the Pfizer/BioNTech or Moderna mRNA vaccines for:
- People age 50 and older, who have received 3 doses, and at least 4 months after the last dose, or
- People 12 or older with “certain kinds of immunocompromise” and at least 4 months since previous dose can get Pfizer only, or
- People 18 or older with the “same certain kinds of immunocompromise” can get another dose of Moderna only.
Basically, everybody over 50, those over 12 or 18 with immunocompromise, and Pfizer getting the nod for the 12-18 group.
That’s… interestingly specific. We know that Pfizer applied for a booster for elders only, whereas Moderna applied for a boost for everybody over 18. The FDA decided on this instead, giving Pfizer more leeway with younger patients. It’s probably because the dose level in Pifzer is lower, and maybe more easily tolerated by smaller bodies? But it’s sort of the opposite of what the companies applied for.
It’s also slightly surprising and frustrating that this was done without review by the external committees (VRBPAC at the FDA and ACIP at the CDC). But only slightly:
- On the one hand, we all understand the need to move quickly. Having another layer of evaluations when the situation is clear is… not something we need. And these vaccines are indeed well-understood empirically, i.e., where they work and the side-effects. So maybe external review wasn’t strictly necessary.
- On the other hand – and I’m just venting my personal frustration here – without the public meetings there’s no public release of documents. We don’t get to see the submission dossier, the slide decks of the various evaluating bodies, and so on. Those were very rich with detail when the vaccines were originally submitted, so I kind of miss them here. It’s possible to get some of the data, but only based on rumor as to what was used, and it takes a lot of digging.
So… ok. The data nerd in me is a little frustrated. And even I admit that the public peer review and critique can sometimes resemble a public flogging; it demonstrates committment for sure, but rather more painfully than necessary.
We’re happy with the end decision to authorize another booster, since there seems to be relatively little occurrence of “antigenic original sin”, and the Omicron-specific vaccines don’t seem to be that much better (so far; we’re not done yet, in that regard). We’ll dig a little for data, but right now this all seems more or less ok.
Zvi at Don’t Worry About the Vase is a bit more negative about the usual peer review meetings, as we would expect. He also quotes Vincent Racaniello, professor of microbiology and immunology at Columbia, on the Israeli data that (apparently) went into the FDA/CDC decision. After complaining that the boosted and unboosted cohorts were not matched for co-morbidities, Racaniello goes into the numbers:
Among participants aged 60 to 69, death from Covid-19 occurred in 5 of 111,776 participants in the second-booster group and 32 of 123,786 participants in the rst-booster group (adjusted hazard ratio, 0.16; 95% CI, 0.06 to 0.41; P<0.001) (Table S2).
Among participants aged 70 to 79, death from Covid-19 occurred in 22 of 134,656 participants in the second-booster group and 51 of 74,717 participants in the rst-booster group (adjusted hazard ratio, 0.28; 95% CI, 0.17 to 0.46; P<0.001) (Table S3).
Among participants aged 80 to 100, death from Covid-19 occurred in 65 of 82,165 participants in the second-booster group and 149 of 36,365 participants in the rst-booster group (adjusted hazard ratio, 0.20; 95% CI, 0.15 to 0.27; P<0.001) (Table S4).
Really, 5 vs 32, 22 vs 51, 65 vs 149 and you are making policy for the US based on this? This is insanity.
(NB: “rst-booster” above should apparently be “first-booster”.)
I’m not as negative as Racaniello here, despite his being very much a big cheese and me being very much a très p’tit fromage. However:
- These effects are all statistically significant by the hazard ratio test (as shown by the $p$-values). So the effect is probably real, i.e., it would reproduce in another experiment.
-
The strength of effect is large enough to be meaningful as well, reducing the death rates by large fold ratios.
For example, in the first group of 60-69, the fold ratio is:
\[\frac{32/123,786}{5/111,776} = 5.78\]
When you get an effect which is both statistically significant and has a good strength of effect, it’s usually time to pay attention.
Now, if I may interpret Racaniello a bit, he seems to be making the point that this intervention successfully reduces a risk, but it was a risk that was already small to begin with. Maybe your time would have been better spent reducing smoking, or treating obesity, or getting gun safety practices more widespread? (I’ve got a list somewhere here, of all the usual liberal interventions I want.)
That might be. But this looks like a pretty low cost & sure thing to me, as a person in that 60-69 age group, so I’m disposed to take it.
Zvi continues, observing that he will pass on the 2nd booster:
That is also because I am young and healthy, so much so that I am not even currently eligible. If I was sufficiently old and/or unhealthy, I would have a lower threshold for boosting, but I would still wait until conditions were getting worse to better time the benefits.
…
- Cost of second booster is small.
- Benefit of second booster is small and temporary.
- If you’re at very high risk, maybe it makes sense.
- Either decision is at worst a small mistake.
Probably skipping is reasonable for Zvi, especially since he’s young & ineligible anyway.
I’m skeptical that one can see a wave coming in advance enough to time a booster. I’m even more skeptical that other people won’t dogpile on the booster supply, making appointments impossible to get when the time comes. So while I’m older but in reasonable health, I want the sure thing of having the booster now while the Omicron/BA.2 wave comes along. I’ll take that 5.78-fold reduction in risk of death for my age decile, thank you very much.
But I agree with Zvi that, given the low risk level for someone with 3 doses already, it’s a marginal decision. Even if the decision (2nd boost vs no 2nd boost) turns out wrong, it’s probably a small error either way, given what we know now.
The General Media


 The general media reported this all over the place; as a representative example here’s
the Reuters coverage. [3] Alas, they omit or mangle most of
the details of who’s eligible when immunocompromised, what the immunocompromises are,
which vaccines go to which age groups, and so on. They got the general idea of “2nd
boosters for the over 50 crowd” right, but pretty much stopped there. That was typical of
most of the general media coverage I read.
The general media reported this all over the place; as a representative example here’s
the Reuters coverage. [3] Alas, they omit or mangle most of
the details of who’s eligible when immunocompromised, what the immunocompromises are,
which vaccines go to which age groups, and so on. They got the general idea of “2nd
boosters for the over 50 crowd” right, but pretty much stopped there. That was typical of
most of the general media coverage I read.
Reuters did point out correctly that given the Republicans blocking any pandemic spending, the government only has money to cover boosters for elders. That is something that will have to be addressed, but it is, alas, not a scientific problem.
STAT News had some slightly more detailed coverage. [4] (Though, amusingly enough, their hyperlink to “underlying medical conditions” lead to apple.com. I mean, yeah, it’s an addiction, but not that kind of addiction!) They noted the lack of VRBPAC and ACIP meetings will attract critics, and that the final age ranges are kind of opposite what the companies applied for. They claimed Moderna applied for a larger age range to give the FDA “flexibility” in assigning age ranges, which I’d never heard before (and view skeptically).
They also correctly, in my view, assess the risk of the oncoming Omicron/BA.2 wave as being a good reason to boost now, though it might leave us more vulnerable come winter. Kind of hard to say which way to dodge, here.
The venerable Boston Globe offers more or less standard coverage. [5]
Interestingly, the venerable Globe quotes some different Israeli data (as usual, mangled so that we don’t know the age cohort beyond “over 60”, let alone know the source), showing 92/328,000 deaths in the group with 4 doses vs 232/234,000 deaths in the group with 3 doses. Even just a simple (as in naïve) statistical test of proportion shows that this is wildly statistically significant, with a 3.5-fold reduction in death rates:
> prop.test(x = c(92, 232), n = c(328000, 234000))
2-sample test for equality of proportions with continuity correction
data: c(92, 232) out of c(328000, 234000)
X-squared = 118.58, df = 1, p-value < 2.2e-16
alternative hypothesis: two.sided
95 percent confidence interval:
-0.0008544266 -0.0005675037
sample estimates:
prop 1 prop 2
0.0002804878 0.0009914530
> 0.0009914530 / 0.0002804878
[1] 3.534746
Though, to take a point from Racaniello above, it is a reduction in a death rate that’s already low. We could do more statistical tests, but the point should be reasonably clear: the effect is real and large, but you can argue about whether it’s worth the bother given the already-low death risk.
The venerable Globe also reports on a number of Boston physicians questioning the timing of boosting, given the unknowns about when another wave might arrive. [6] Yes, we can’t do it perfectly with exact predictions of the future. Zvi thinks he can see the wave coming in time to get boosted (see above), but I’d rather get boosted now while I see Omicron/BA.2 coming. There’s no way to do this perfectly, so no sense complaining.
Ok, So Should I Get Boosted Again, or Not?
 So given all that, there’s no shortage of people wanting advice on whether or not to get
another booster. Thus there is no shortage of media articles offering said advice. One such is
from Apoorva Mandavilli in the New York Times [7]:
So given all that, there’s no shortage of people wanting advice on whether or not to get
another booster. Thus there is no shortage of media articles offering said advice. One such is
from Apoorva Mandavilli in the New York Times [7]:
- She summarizes the situation, including the woefully low acceptance rate of first boosters so far in the US.
- Then she notes that there’s not exactly a scientific consensus on 2nd boosters. The Israeli study on which it’s based was posted only last week, and is a preprint, i.e., not yet peer reviewed. [11] (I also haven’t waded through it personally yet, though there’s no especial reason you should care about that. We’ll consult Eric Topol later on this subject, anyway.)
- The Israeli study is “decisive” in terms of mortality rate, but flawed: all the people who got the 4th shot were volunteers (i.e., not randomized), and thus the 3-shot and 4-shot cohorts are sort of compromised statistically. Mandavilli’s explanation here was pretty nice.
- She quotes Robert Wachter of the Department of Medicine at UCSF:
As a healthy 64-year-old man whose third shot was seven months ago, I will get one this week if I can.
Sounds good enough for me, as a reasonably healthy mid-60s type. (But in true both-sider fashion, Mandavilli goes on to quote other experts with other opinions.)
- And, of course, we don’t understand quantitatively the effects of prior COVID-19 disease, or antigenic original sin.
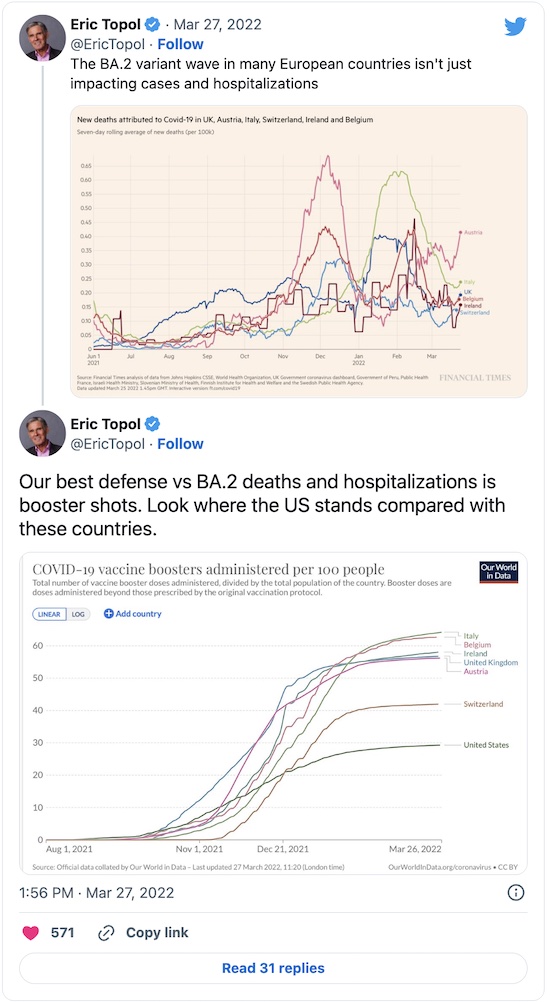
Next, lets consult Eric Topol of the Scripps Translational Research Institute. He’s had a few things to say on Twitter, as well as on his blog Ground Truths:
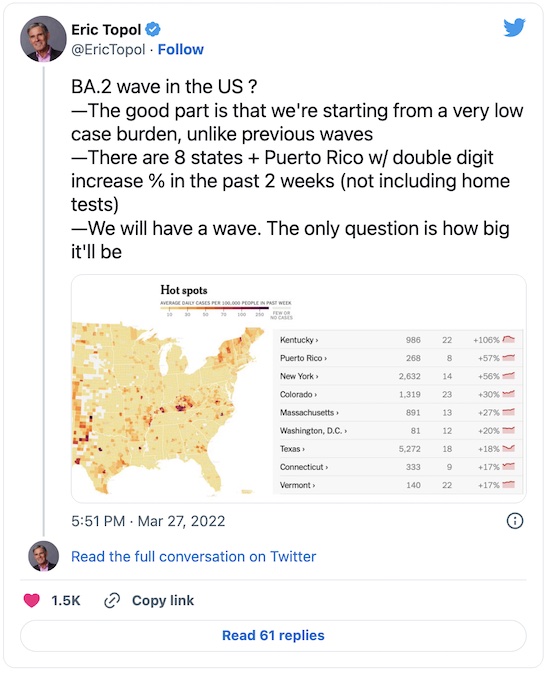
First, there is definitely an Omicron/BA.2 wave coming; the only question is how big:
Second, there’s some evidence that mixing the Pfizer/BioNTech and Moderna vaccines as mutual boosters gives a salubrious result:
Third, the US is woefully under-boosted. Compare the US to European countries, and then think about how the Omicron/BA.2 wave is hitting them vs how it will hit us:
Consulting Experts in Longer Form
Next, let’s consult Topol in longer form at his blog Ground Truths [8], and the epidemiologist Katelyn Jetelina at her blog, Your Local Epidemiologist. [9] They’re both well worth your time. Really.
Eric Topol @ Ground Truths
 Topol’s opener summarizes the question on the minds of everybody who’s paying attention:
Topol’s opener summarizes the question on the minds of everybody who’s paying attention:
The inconvenient truth is that we are going to experience a new BA.2 variant wave in the United States—the magnitude of which remains uncertain—and, this highlights the question of whether a 2nd booster (4th shot) would be useful.
There will be an Omicron/BA.2 wave. How bad is uncertain, but we know that boosters will help. How much will they help, and should you get one?
One thing that will help is previous Omicron/BA.1 or Omicron/BA1.1 infection: there should be at least some cross-immunity. Since estimates now say almost 40% of Americans got the first wave of Omicron, they should have some protection against BA.2.
But the other thing that will help even better is a recent booster. Topol shows a summary table from the preprint of a Qatari study [12]
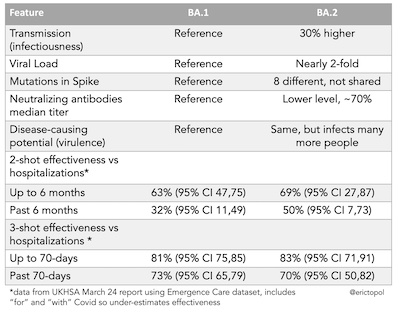 Look at the last 4 rows there: 3-shot efficacy vs hospitalization is 70% - 80%. A recent fourth
shot in a BA.2 wave would be even better.
Look at the last 4 rows there: 3-shot efficacy vs hospitalization is 70% - 80%. A recent fourth
shot in a BA.2 wave would be even better.
In fact, the efficacy of a 2nd booster has been studied already a couple of times, all during the Israeli Omicron wave. [13] [14] [15]
They range from a small safety study of health-care workers, to a 1 million+ person study
comparing 3rd vs 4th shots. Their results are quite strikingly in favor of another
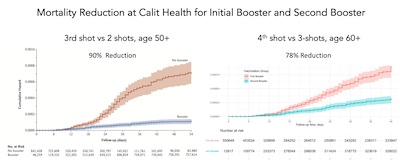
booster. For example, the Kaplan-Meier curves for 3 shots vs 2 shots (1st booster vs Delta)
and 4 shots vs 3 shots (2nd booster vs Omicron) are pretty similar, showing almost as much of a
boost from the second booster as from the first:
 Now, there are of course several serious issues here, the first couple of which are as
pointed out above by Racaniello to Zvi:
Now, there are of course several serious issues here, the first couple of which are as
pointed out above by Racaniello to Zvi:
- The populations were not especially well matched (though they were partially matched). More to the point, those receiving the 4th shot were volunteers, not randomized! This means the study is convolved with a confounder that will overstate the effect, as volunteers are likely more COVID-cautious in the first place.
- The base rate is low: we’re talking again about a statistically significant and strong effect of reduction of death, but for a death rate that was already small. It may, however, be important at the population level: all those elder deaths eventually add up!
- The plot on the left was during Delta, while the plot on the right is during Omicron. This is likely why the effect is slightly less dramatic for Omicron: 78% reduction in hospitalization vs 90% reduction. Omicron is nasty.
So those are about the same cautions we teased out above.
Topol also points out the unpredictability of SARS-CoV2, and how unlikely we are to intuit the onset of a wave in time to get boosted (emphasis in original):
It is noteworthy that several countries like India, Bangladesh, and Sweden transitioned to ~100% BA.2 without experiencing a new case increase. Yes, this virus is as unpredictable as it is formidable.
Topol’s summary recommendation is in line with the decision toward which I was already leaning:
I would recommend the 2nd booster if you are more than 4-6 months from your 3rd shot, you are age 50+, you tolerated the previous shots well, and you are concerned about the BA.2 wave where you live, or that it’s getting legs as you are trying to decide. Or if you are traveling or have plans that would put you at increased risk.
So for me, yeah… boost. Though he does point out that if you’ve had 3 doses of vaccine and caught Omicron anyway (really due to just plain bad luck), you can skip the second booster. Or not, since the 2nd booster is low cost and low risk.
Katelyn Jetelina @ Your Local Epidemiologist
 Jetelina opens by pointing out that fourth doses of mRNA vaccines have been underway not
just in Israel, but also in the UK and Germany. So this is by no means unusual. The US
is, as regrettably usual, playing catch-up.
Jetelina opens by pointing out that fourth doses of mRNA vaccines have been underway not
just in Israel, but also in the UK and Germany. So this is by no means unusual. The US
is, as regrettably usual, playing catch-up.
Oddly, the FDA VRBPAC committee meeting next week was to debate second boosters, but specifically was told there would be no votes on any particular application. Since the FDA approved those applications this week, we now see why that caveat was there. Still… pretty odd, though.
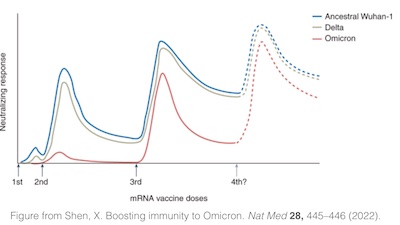
- A paper by Shen in Nature Medicine [16] points out that
both empirical evidence from Europe
and quantitative models agree that a 3rd dose against Omicron is about as effective as a
second dose against Delta. In other words, Omicron is nastier and another booster seems
altogether in order.
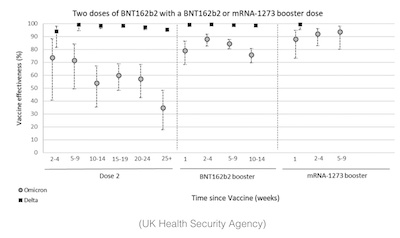
- The UK Health Services Agency reports [17] that hospitalization efficacy vs both Delta and Omicron are holding up, better than efficacy vs any infection at all. But Omicron is worse, so again a booster seems in order to cope with Omicron.
- Moderna holding up a bit better, compared to Pfizer/BioNTech. [18]
But again, Topol’s citation of Kaplonke above indicates combination of vaccines, so I’m
inclined on that basis and on this point from Jetelina to get a Moderna booster on top
of my Pfizer doses.
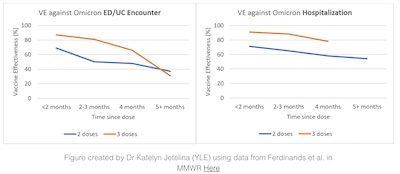
- US vaccine efficacy seems to be declining slightly, as shown in this graph by Jetelina from the CDC’s MMWR data. [19]
- Interestingly, ref13 from Regev-Yochay (one of the Israeli studies cited above) notes that immunogenicity in younger health care workers maxes out @ 3 doses. So for younger folks there may be no point in 4th dose/2nd booster.
So Jetelina gives no specific advice, but seems broadly consistent with Topol.
Of Course There Will be Problems…
 As long as Republicans can throw a wrench into the works, there will be problems as
documented today by Leonhardt at the NYT. [10]
Basically, we face a number of problems here:
As long as Republicans can throw a wrench into the works, there will be problems as
documented today by Leonhardt at the NYT. [10]
Basically, we face a number of problems here:
- In the short term, too few people are seeking vaccination. That includes primary vaccination, boosters, and now second boosters. The graph above shown in Topol’s tweet shows the US far behind many European countries… the countries currently being trounced by Omicron/BA.2. How much more badly will the US get trounced, with a lower vaccination level overall, and no effective universal health care?
- In the long term, we haven’t bought enough doses of vaccine. Partly that’s because of congressional obstinacy, with Republicans wanting to claw back money from the states to pay for it. Given the razor’s edge of most state budgets, that is just impossible. So if there’s a surge, we’ll need more vaccine doses, but we won’t have the money to buy them and most of them will be bought by other countries anyway.
The sheer predictability of all this is the most tiresome part. Can’t we elect fewer Republicans and start doing useful things again?
The Weekend Conclusion
Well, this was a bit of a lengthy trawl through the media and bits of the scientific literature, wasn’t it? Still, I think it led to a pretty firm conclusion for someone of my age and COVID caution level. (Younger, less risk-averse people will of course reach the opposite conclusion.)
It looked to me, taking advice from Wachter & Topol’s quote of the Kaplonke paper in Science, as a past-mid-60s guy whose last shot was 5 months ago, that I should take the 2nd booster. And it should be Moderna, to offset my previous 3 doses of Pfizer.
A bit of poking around on the day after the FDA approval revealed that nobody was prepared to provide it yet (sigh). Poking around the next day revealed that all slots were taken, for that day and the next (sigh). (This was starting to remind me of the spring of 2021, with the vaccine chaos.) Still, I managed to snag a Moderna appointment on Friday (today).
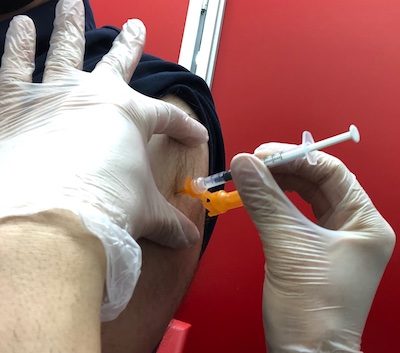
This afternoon I took a short sail in the Weekend Zeppelin for the appointment. (In reality a boring bus ride and train ride.) I arrived at a local vaccine dispensary for my appointment. Here is your humble Weekend Editor’s portside dorsal manipulator tentacle, being injected with a 2nd booster. Moderna, this time. (You know, I wish I could ride in a real zeppelin, or one of the modern airships like the LMH-1. Parking might be more of a problem than usual, though. That would more than offset any vaccine reaction discomfort!)
Tune in tomorrow for an after-action report of the side-effects. Given the higher dose of Moderna, I expect a bit more in the reactogenicity department here, than with Pfizer/BioNTech. But… happily.
Addendun 2022-Apr-02: After-Effects
Really not bad: kind of sore in my deltoid, a little sore and feverish last night, but overall not bad.
I’m… vaguely disappointed.
Addendum 2022-Apr-05: After-After-Effects
Ok, it lasted longer and was a bit more intense than my reaction to Pfizer, but that was expected. It’s only today that I’m finally not completely exhausted.
An acceptable price to pay for better immunity.
Notes & References
1: FDA Office of Media Affairs, “Coronavirus (COVID-19) Update: FDA Authorizes Second Booster Dose of Two COVID-19 Vaccines for Older and Immunocompromised Individuals”, FDA News Release, 2022-Mar-29. ↩
2: CDC Media Relations, “CDC Recommends Additional Boosters for Certain Individuals”, CDC Newsroom Releases, 2022-Mar-29. ↩
3: M Erman & M Maddipatla, “U.S. authorizes second COVID booster for Americans 50 and older”, Reuters, 2022-Mar-29. ↩
4: A Joseph, “U.S. approves second Covid-19 booster for people 50 and older”, STAT News, 2022-Mar-29. ↩
5: L Neergaard & M Perone, “FDA authorizes another booster dose of the Pfizer or Moderna COVID-19 vaccine for people age 50 and up”, Boston Globe, 2022-Mar-29. ↩
6: R Cross, “Some doctors question timing of second COVID booster in absence of surge”, Boston Globe, 2022-Mar-29. ↩
7: A Mandavilli, “Should You Get Another Covid Booster?”, New York Times, 2022-Mar-29. ↩
8: E Topol, “A new wave and a new booster?”, Ground Truths blog, 2022-Mar-29. ↩
9: K Jetelina, “Another mRNA booster or not?”, Your Local Epidemiologist blog, 2022-Mar-25. ↩
10: D Leonhardt, “Two Covid Problems”, New York Times, 2022-Mar-31. ↩
11: R Arbel, et al., “Second Booster Vaccine and Covid-19 Mortality in Adults 60 to 100 Years Old”, preprint at Research Square, posted 2022-Mar-24. ↩
12: HN Altarawneh, et al., “Effect of prior infection, vaccination, and hybrid immunity against symptomatic BA.1 and BA.2 Omicron infections and severe COVID-19 in Qatar”, medRχiv, 2022-Mar-22. ↩
13: G Regev-Yochay, et al., “Efficacy of a Fourth Dose of Covid-19 mRNA Vaccine against Omicron”, New Engl Jnl Med, 2022-Mar-16. DOI: 10.1056/NEJMc2202542. ↩
14: YM Bar-On, et al., “Protection by 4th dose of BNT162b2 against Omicron in Israel”, medRχiv, 2022-Feb-01. ↩
15: R Arbel, et al., “Second Booster Vaccine and Covid-19 Mortality in Adults 60 to 100 Years Old”, preprint for Nature Portfolio at Research Square, 2022-Mar-24. DOI: 10.21203/rs.3.rs-1478439/v1.↩
16: X Shen, “Boosting immujnity to Omicron”, Nature Medicine 28, 445-446, 2022-Feb-24. DOI: 10.1038/s41591-022-01727-0. ↩
17: UK Health Security Agency, “COVID-19 vaccine surveillance report, Week 12 (24 March 2022)”, UK Health Security Agency reports, 2022-Mar-24. ↩
18: R Pajon, et al., “SARS-CoV-2 Omicron Variant Neutralization after mRNA-1273 Booster Vaccination”, New Engl Jnl Med, 386:1088-1091, 2022-Mar-17. DOI: 10.1056/NEJMc2119912. ↩
19: JM Ferdinands, et al., “Waning 2-Dose and 3-Dose Effectiveness of mRNA Vaccines Against COVID-19–Associated Emergency Department and Urgent Care Encounters and Hospitalizations Among Adults During Periods of Delta and Omicron Variant Predominance — VISION Network, 10 States, August 2021–January 2022”, CDC Morbidity and Mortality Weekly Report, 71:7, 255-263, 2022-Feb-18. ↩


Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.