What's happening in the Moderna/Lonza COVID vaccine phase 3 start?
Tagged:COVID
/
PharmaAndBiotech
/
SomebodyAskedMe
Somebody asked me (really it’s the same “somebody” behind all of these) about the Moderna COVID vaccine phase 3 start. Basically, it looks like the start of a reasonably designed Phase 3 trial: the beginning of a long slog to test safety & efficacy.
Data
First, as always, links to the data sources:
| NIH Twitter | NIH Press Release | ClinicalTrials.GOV |
|---|---|---|
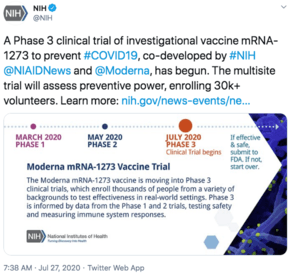 |
 |
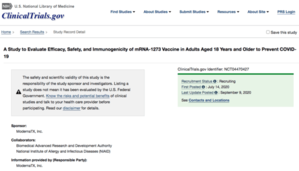 |
How to volunteer: See the good folk at the coronavirus prevention network. Trial sites are all around the US concentrated in cities, one of them my home. Somewhat of a puzzle to me that all sites are in the US, not internationally?! Who even does that nowadays? I imagine it will fill up fast – I asked the Weekend Editrix if we should volunteer, and got a somewhat equivocal response.
Some Details
A few details from reading the clinical trial registry entry:
- ~30k volunteers.
- Complex inclusion/exclusion criteria, but basically over 18 and unlikely to get pregnant. A few things about not taking immunosuppressants, not having had COVID-19, not having had terrible reactions to vaccines in the past, etc. But basically a broad population, including those with relevant pre-existing conditions (as is relevant here in an efficacy trial, no longer a safety trial). Eligibility criteria here on ClinicalTrials.gov.
- 2 injections, on days 1 and 29. Presumably some blood draws, on days of injection and assessment days, but I didn’t read deeply enough to find out how many. (Nope, I take it back – Time Frame: Day 1, Day 29, Day 57, Day 209, Day 394, and Day 759. So not bad – only 6 blood draws. I thought there would be more!)
- Placebo controlled. Placebo is a saline injection. (I’m slightly surprised they don’t give you something to induce mild fever and aches, so you won’t know which arm of the trial you’re in. Because everybody gets aches & pains & fever in Phases 1 & 2. Perhaps a flu vaccine in the control arm? For example, in a trial for ketamine injections to treat depression, where I had a personal interest a couple years ago, the control arm gets an unrelated benzodiazepine, so they’ll feel woozy and not know which arm of the trial they’re in.)
- Quadruple blinded: participant, caregiver, investigator, and data analyst. They’re being verrry careful, which warms the heart of this skeptical, flinty, grizzled old statistician. (Well, not exactly warm-hearted, but slightly less cold than usual.)
- Preliminary readout at end of October 2020, but official monitoring for 759 days (basically 2 years) for absolute final readout (so something like July 2022, but thumbs up/down for at least initial FDA approval likely by end of this year or early next year; apparently Lonza — Moderna’s partner — is already doing at-risk manufacturing mostly in Switzerland).
I’d have been interested in volunteering if the Weekend Editrix had said ok, but for now with “ambiguous” health insurance status at least for a while, maybe it’s not the time for me to be confusing our rather cruel & arbitrary health care delivery system.
Unsurprisingly, as of a couple days after that announcement, there were over 100,000 volunteers for this 30,0000 person trial. They’ll pick a subset to ensure diversity over pre-existing condition, age, body mass index, gender, race, and so on. Probably little chance to get in, should you still be interested at this point.
Multiply-Blinded, Placebo Controlled Design
What’s remarkable is the number of times when designs are not thoughtful, when the MDs running the trial think they have no need of a statistician to design the experiment. (Of course, I’m a grizzled old statistician, so I would say that, wouldn’t I?)
For example, this trial – NCT01920555 – looks at the use of ketamine for rapid treatment of major depressive disorder. I have both a personal interest in depression, and I also know some of the people involved (mainly the research anesthesiologist who had to administer the doses was a good friend, so alas, I couldn’t volunteer). But to make sure subjects couldn’t tell which arm of the trial they were in, they gave ketamine to the treatment arm and midazolam (unrelated benzodiazepine) to the control arm. Everybody gets to feel a little woozy for 30min or so, and the average person off the streets can’t tell the difference. (Probably an anesthesiologist could tell the difference, as could a ketamine abuser, but they wouldn’t recruit such a subject.)
This was an example of a good design.
An earlier trial of ketamine – NCT00088699 – was a mediocre design: the placebo was “an inactive substance”, probably saline. So subjects could probably tell the difference, if they knew what ketamine was (and anybody in a trial would probably look it up). They tried to compensate with a crossover design, where at the second dose they swapped treatment and control arms – so every subject got 1 treatment and 1 control dose. But by the second dose, I bet the subjects could definitely tell which was which!
This sort of thing makes research statisticians sad. But it very definitely doesn’t seem to be a pitfall with the Moderna/Lonza COVID trial. So good on them.

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.