How Can Evusheld Possibly Work?!
Tagged:COVID
/
JournalClub
/
MathInTheNews
/
PharmaAndBiotech
/
Statistics
Evusheld is an antibody infusion which confers about 6 months of vaccine-like immunity to COVID-19. How can that possibly work, when antibodies last a few days to a few weeks?!
Evusheld
 Evusheld was approved last December, as we wrote at the time. [1]
Interestingly, it was approved directly by the FDA, bypassing the usual VRBPAC committee
of external experts for review.
Evusheld was approved last December, as we wrote at the time. [1]
Interestingly, it was approved directly by the FDA, bypassing the usual VRBPAC committee
of external experts for review.
It’s 2 antibodies (tixagevimab and cigavimab) given as simultaneous injections. They target distinct areas of the SARS-CoV2 spike protein.
The interesting bit: This confers immunity at efficacy levels of 77% for 6 months, starting more or less immediately!
Now, if you’re an immunocompromised individual who cannot medically tolerate vaccination (2% - 3% of the US population), this is a big deal, even if it requires dosing twice a year forever, at a cost of $1,000/dose.
Vaccination is about $30/dose, for comparison.
But the puzzle to me was: given that infused antibodies last somewhere from days to weeks in your body since you’re not making them, how can you get immunity for 6 months? Today we’ll dive into that question, and get a (partial) answer.
The evidence it works
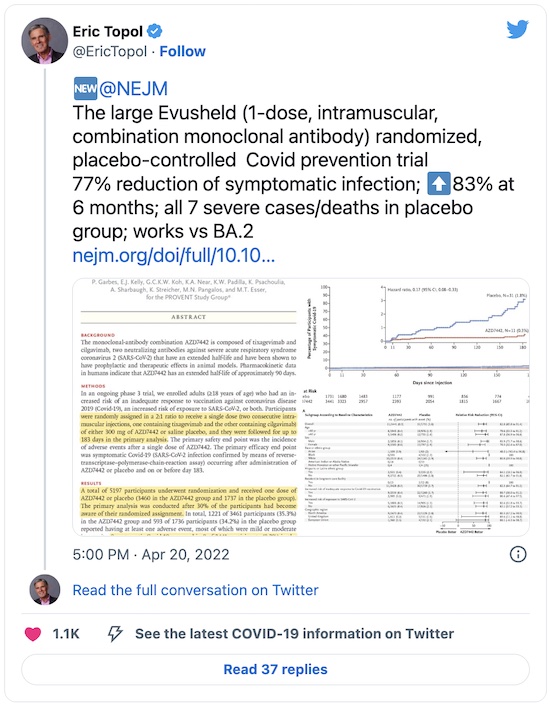
First, Eric Topol of Scripps tells us it seems to work pretty well:
By way of evidence, Topol points us at 2 articles in the New England Journal of Medicine [2] and Science Translational Medicine [3], both top-shelf sources for this sort of thing.
 First, let’s have a look through the NEJM paper, which is the publication of the clinical
trial results for Evusheld.
First, let’s have a look through the NEJM paper, which is the publication of the clinical
trial results for Evusheld.
We’ll check their numbers for efficacy using our kludgey little binomial confidence interval R script. [4] They used a much more sophisticated Poisson regression method with robust variance, but didn’t publish enough details for us to reproduce that exactly. So we’ll just be looking for crude agreement with our crude method, nothing more.
They report that their 2 antibodies were from the B cells of convalescent patients, with some modifications discussed below. Also very interestingly, they dose these antibodies at 6x - 22x higher than the levels found in convalescent patients! We’ll see why below.
First up, they assessed mild to moderate adverse events. Keep in mind that by saying “mild to moderate”, they’re counting even if people just report a headache, a cold, or something minor like that to their doctor. They saw such mild events in:
- 1221/3461 patients in the treatment group, or 35.3%
- 593/1736 patients in the control group, or 34.2%
We shouldn’t expect that to be statistically significant, and indeed it’s not; the efficacy against mild/moderate adverse events was broadly consistent with 0%:
> efficacyAndCL(3461, 1221, 1736, 593)
LCL Eff UCL
-0.11874715 -0.03278303 0.04547047
The treatment and control groups are pretty much identical as far as mild/moderate adverse events go. That’s good; at least evusheld doesn’t make anything worse.
Next, let’s look at symptomatic COVID-19 and death rates.
At an early readout, when 30% of the patients had discovered the arm the trial to which they’d been assigned, they report:
- 8/3441 patients in the treatment group
- 17/1731 patients in the control group
They report an efficacy of 76.7% (95% CL: 46.0% - 90.0%). We broadly agree with that, rounded to 2 significant figures:
> efficacyAndCL(3441, 8, 1731, 17)
LCL Eff UCL
0.4649044 0.7632699 0.8952743
So I’m not worried about their efficacy as much as I’m worried how 30% of the patients somehow discovered which arm of the blinded trial in which they were enrolled!
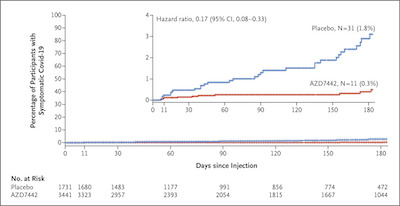 The Kaplan-Meier curve, shown here, gives impressive results in terms of the hazard
ratio. The blue curve is the control group, and we see cases rising; the red curve is the
treatment group where cases rise much more slowly. They reported results on extended
follow-up at 6 months of 82.8% (95% CL 65.8% - 91.4%). Taking numbers from the KM plot,
we broadly agree, again to a couple significant figures with our crude methods:
The Kaplan-Meier curve, shown here, gives impressive results in terms of the hazard
ratio. The blue curve is the control group, and we see cases rising; the red curve is the
treatment group where cases rise much more slowly. They reported results on extended
follow-up at 6 months of 82.8% (95% CL 65.8% - 91.4%). Taking numbers from the KM plot,
we broadly agree, again to a couple significant figures with our crude methods:
> efficacyAndCL(3441, 11, 1731, 31)
LCL Eff UCL
0.6502766 0.8214979 0.9089119
There were 5 deaths, all in the control group. While they didn’t report an efficacy here, we can crudely estimate it as 100% (95% CL: 61.4% - 100%):
> efficacyAndCL(3441, 0, 1731, 5)
LCL Eff UCL
0.6137538 1.0000000 1.0000000
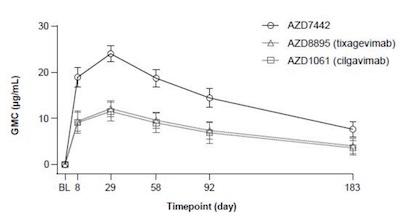 But do the antibodies actually stick around for 6 months? That’s an astonishingly long
time, since the normal half-life is days to a week or so.
But do the antibodies actually stick around for 6 months? That’s an astonishingly long
time, since the normal half-life is days to a week or so.
The answer is yes, as seen in the pharmacokinetics curve shown here (Supplement figure 2A in the paper). It looks like the geometric mean concentration (GMC) goes down by a factor of about 2.5, maybe a bit more.
- Elsewhere they make the claim that the half-life is 3 months, so we’d expect them to go down by a factor of 4, so apparently the observational data is even better than that.
- But if you dose 6x - 22x higher than the clinically meaningful level found in convalescent patients, then after 2 half-lives those should go down by a factor of 4.
In other words, you’d expect the concentration to remain above the level found in convalescents for at least 6 months.
So there’s a partial answer to our question: engineer the antibody to have a longer half-life, and then dose it really high so after a couple half-lives it’s still at an effective level. Sophisticated antibody engineering and brute-force dosing.
 The second paper covers rather similar ground, so I won’t drag you through all of it.
The second paper covers rather similar ground, so I won’t drag you through all of it.
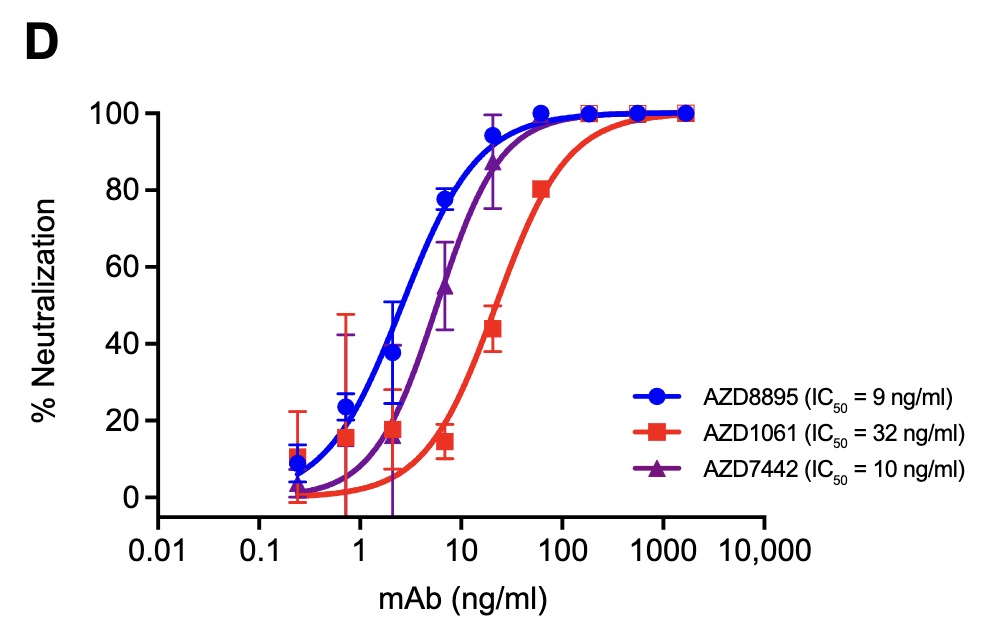
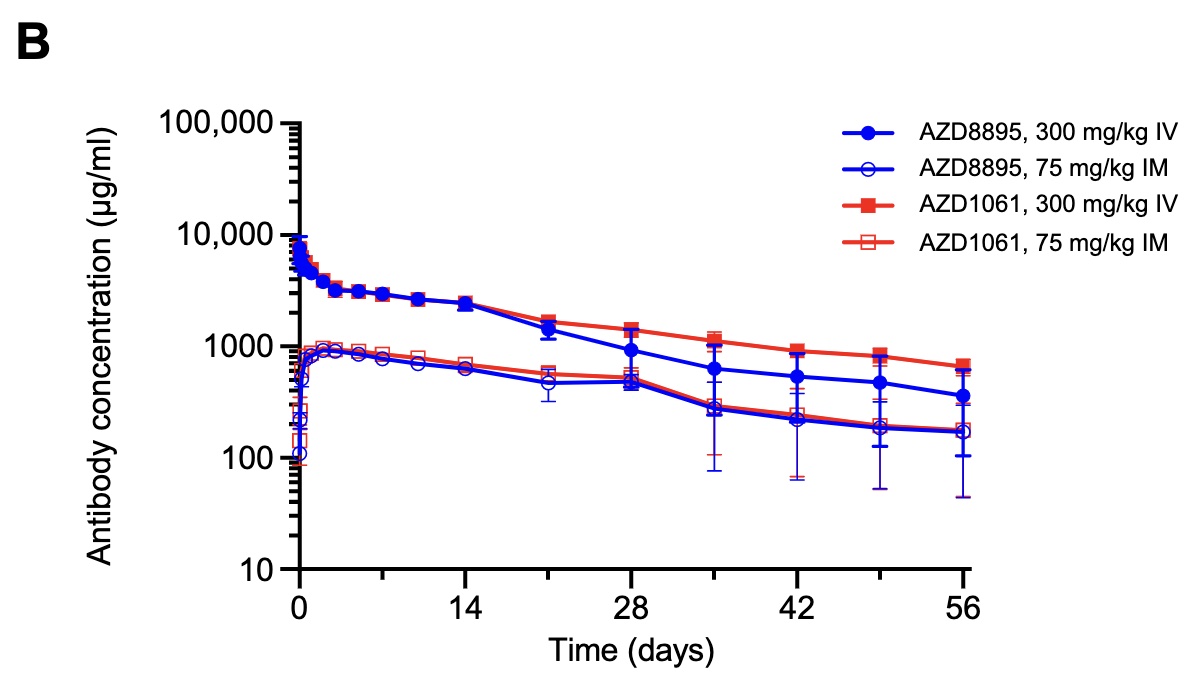 But there are 2 particular sub-figures I want to highlight, which tell us (a) what
antibody levels seem to be sufficient to neutralize the virus, and (b) how long antibody
levels can be kept above that. These are Figures 1D and 2B in the paper, shown here.
But there are 2 particular sub-figures I want to highlight, which tell us (a) what
antibody levels seem to be sufficient to neutralize the virus, and (b) how long antibody
levels can be kept above that. These are Figures 1D and 2B in the paper, shown here.
- Figure 1D tells us that the EC50 (concentration at which you get half the maximum effect) is 10 - 30 ng/ml, depending on which antibody you’re discussing. But by the time you get up to 1000 ng/ml = 1μg/ml you’re getting a full kill. That’s a level high enough to eliminate the virus, more or less completely, if humans can stand that level.
- Figure 2B tells us the lifetime of these antibodies. It looks like the initial doses were around 1,000 - 10,000 μg/ml, and declined to 500 - 1,000 μg/ml after 2 months. That is, absolutely monstrously effective levels (starting at 1000x - 10,000x the 100% kill dose seen above!) were sustained for months.
Now I can begin to see that it makes sense that this thing works: very high doses are well tolerated, and the half-life is long enough to give 6 months of immunity.
Antibody engineering


 But… but… but! How exactly did they get the antibodies to have such
amazingly long half-lives?! Following the references in their bibliographies led us to 3
papers (actually more, I just ran out of energy at 3) from 2002 - 2013 on this
subject. [5] [6] [7]
But… but… but! How exactly did they get the antibodies to have such
amazingly long half-lives?! Following the references in their bibliographies led us to 3
papers (actually more, I just ran out of energy at 3) from 2002 - 2013 on this
subject. [5] [6] [7]
It appears there’s a thing called the “YTE substitution”, which in the usual protein mutation notation makes the triple substitution M252Y/S254T/T256E (hence YTE). This was found to extend dramatically the half-life of antibodies, by 2-4x. Since these are somewhat older papers, they were of course done with antibodies that had nothing to do with SARS-CoV2. The discovery with Evusheld was that the technique carried over to SARS-CoV2.
Everybody points back to the Dall’Acqua papers as the source of this magic. But a quick read-through was maddening: although the results were described, they were not motivated. How in the world did anybody think to try that?! I’m mystified.
The Weekend Conclusion… and Some Remaining Puzzles
So the answer to our question of “how can it possibly work?!” seems to be, at the surface level:
- The YTE modification is known to increase antibody half-life in the body, in this case around 90 days.
- They administer it at very high levels, maybe 6x - 22x what is required, so after 2 half-lives (180 days) it’s still well above prevention-effective levels.
But there are still some questions that bother me:
- How was the YTE substitution discovered? They weren’t just messing about by chance!
- How does the YTE substitution work? What exactly is the physics of why antibodies with this triple substitution live so long?
- Why isn’t this substitution done for every antibody infusion? For example, if we take bebtelovimab, the antibody infusion still in use to treat COVID-19 Omicron, would it be better if we did the YTE substitution to it?
So we learned something… and we also learned how little we know.
A typical day in science.
Notes & References
1: Weekend Editor, “FDA Grants EUA to AstraZeneca’s Evusheld for COVID-19 Prevention”, Some Weekend Reading blog, 2021-Dec-09. ↩
2: MJ Levin, et al., “Intramuscular AZD7442 (Tixagevimab–Cilgavimab) for Prevention of Covid-19”, N Engl Jnl Med, 2022-Apr-20. DOI: 10.1056/NEJMoa2116620. ↩
3: Y-M Loo, et al., “The SARS-CoV-2 monoclonal antibody combination, AZD7442, is protective in nonhuman primates and has an extended half-life inhumans”, Sci Transl Med 14, eabl8124, 2022-Mar-09. DOI: 10.1126/scitranslmed.abl8124. ↩
4: Weekend Editor, “Script to asses efficacy and 95% CL using binomial confidence intervals”, Some Weekend Reading blog, 2021-Nov-12. ↩
5: GJ Robbie, et al., “A Novel Investigational Fc-Modified Humanized Monoclonal Antibody, Motavizumab-YTE, Has an Extended Half-Life in Healthy Adults”, Antimicrob Agents Chemother 57:12, 6147-6153, 2013-Dec. DOI: 10.1128/AAC.01285-13. ↩
6: WF Dall’Acqua, et al., “Properties of Human IgG1s Engineered for Enhanced Binding to the Neonatal Fc Receptor (FcRn)”, Jnl Biol Chem 281:33, 23514-23524, 2006-Aug-18. DOI: 10.1074/jbc.M604292200. ↩
7: WF Dall’Acqua, et al., “Increasing the Affinity of a Human IgG1 for the Neonatal Fc Receptor: Biological Consequences”, Jnl Immunol 169:9, 5171-5180, 2002-Nov-01. DOI: 10.4049/jimmunol.169.9.5171. ↩

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.