Statins & ACE inhibitors & ARB blockers as COVID-19 therapeutics?
Tagged:COVID
/
JournalClub
/
R
/
SomebodyAskedMe
/
Statistics
Somebody asked me (honest!) about a news item linking improved treatment outcomes for COVID-19 with patients using statin and ACE inhibitor drugs. Very preliminary stuff, and the effect size is not giant, but it is peer-reviewed and looks quite legit. Not gonna change the world, but even small bits of good news are welcome.
Initially I was a bit suspicious:[1] [2] there’s some evidence that ACE inhibitors actually increase the expression of ACE2, which SARS-COV-2 uses to enter cells, and that would increase risk. Also, the paper is by a bunch of cardiologists, apparently trying to tell us their discipline rules the world. Finally, it was a press release (reference 1), and your Weekend Editor has a rather dyspeptic view of press releases.
Happily, I was wrong on all counts. This looks like preliminary but legit evidence (reference 2) of at least some lowering of the risk of mild COVID becoming severe. There was an accompanying paper (reference 3) on the mechanism, involving 25-hydroxy-cholesterol and several effect-rescue assays that also look pretty good. They’ve got clinical evidence of effect, and lab bench evidence of a mechanism of action. Now they need confirmation from an independent lab, maybe a multi-center test, and they’ll be ready to figure out how to use the information in combat situations.
Clinical effect of statins on COVID-19
 First, let’s take a look at Daniels, et al. (reference 2) and their report on statins
(to lower cholesterol), ACE inhibitors (to lower blood pressure), and ARB blockers (also
to lower blood pressure by a related mechanism):
First, let’s take a look at Daniels, et al. (reference 2) and their report on statins
(to lower cholesterol), ACE inhibitors (to lower blood pressure), and ARB blockers (also
to lower blood pressure by a related mechanism):
- They wondered if statins, ACE inhibitors, and ARB blockers interacted with COVID-19, given they are all but ubiquitous in middle-aged and older people (in developed countries who have access to health insurance, both of which are sometimes doubtful in the US). Also, see reference 4 for other speculation about cholesterol, bp, and COVID.
- They did a single-center study, at UCSD Health. This is a bit of a weak point, which should be addressed in a multi-center study to rule out any oddities associated with UCSD. But it’s a start.
- They looked at 170 patients hospitalized for COVID-19, and 5281 COVID-negative controls. They measured time from hospital admission to onset of severe disease (death or ICU), time to discharge with no longer severe disease, or time to death.
- They did multivariate logistic regressions (in R, bless their little hearts) to account for the effect of statin/ACE/ARB drugs, and to account for confounding factors like age, sex, obesity, etc. [3]
- Statins were strongly associated with reduced risk of progressing to severe disease and with faster recovery times, by a couple of interesting statistical tests (q.v.).
- But… statin use in the COVID-negative controls was more weakly associated with progression: it won’t stop you getting COVID-19, but it helps (a little) once you already have it.
- They say there is “potential evidence” associating ACE/ARB drugs to faster recovery, but their statistical significance tests say otherwise. No idea why they said “potential evidence”, when it’s clear their statistician told them “No.” More medically important, though, is the implicit result: ACE/ARB drugs don’t help against COVID, but they also don’t hurt, so if you’re taking them already for blood pressure you should continue to take them as your doctor prescribes.
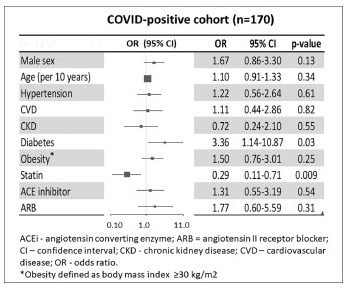 Here’s a summary of their results. They’re measuring the odds ratio of progression to
severe COVID-19 (and recall that, as they’re using logistic regression, we’re always
dealing in log odds ratios). The predictors are in the column on the left. The thing in the
middle is sometimes called a “forest plot”: the favorable situation (odds ratio < 1.0) is on
the left, and unfavorable on the right. You want to see the central blob and its 95%
confidence interval be on one side or the other of 1.0 in order to declare that variable
is a good predictor. Note that: use of statins is a good predictor (and in the good
direction); diabetes is a good predictor (in the bad direction); nothing else makes the
grade (including ACE/ARB drugs).
Here’s a summary of their results. They’re measuring the odds ratio of progression to
severe COVID-19 (and recall that, as they’re using logistic regression, we’re always
dealing in log odds ratios). The predictors are in the column on the left. The thing in the
middle is sometimes called a “forest plot”: the favorable situation (odds ratio < 1.0) is on
the left, and unfavorable on the right. You want to see the central blob and its 95%
confidence interval be on one side or the other of 1.0 in order to declare that variable
is a good predictor. Note that: use of statins is a good predictor (and in the good
direction); diabetes is a good predictor (in the bad direction); nothing else makes the
grade (including ACE/ARB drugs).
So that was time to progression to severe disease. They also did a similar analysis for time to death. However, since nothing was statistically significant, it was exiled to the supplement of the paper. The supplements are always either (a) where the good stuff is, in detail, or (b) where the nonsignificant data goes to die.
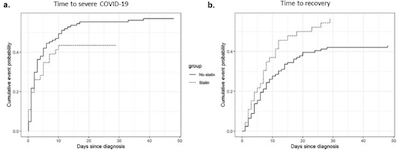 They did something called a Cox proportional hazards model to (essentially) measure
probability per unit time to the onset of severe disease, or to recovery based on the
statin/non-statin cohorts. From those, you make Kaplan-Meier plots (conventionally flipped
vertically compared to here), showing the cumulative progressions/recoveries vs time. By comparing the
black line with the dotted line, you can see that the statin patients were slower (or
never) to progress to severe disease, and faster to recover. More quantitatively, they
computed the usual hazard ratio and associated logrank $p$-value, to assess whether the curves are
statistically significantly different, not just visually. From table 2 in the paper:
They did something called a Cox proportional hazards model to (essentially) measure
probability per unit time to the onset of severe disease, or to recovery based on the
statin/non-statin cohorts. From those, you make Kaplan-Meier plots (conventionally flipped
vertically compared to here), showing the cumulative progressions/recoveries vs time. By comparing the
black line with the dotted line, you can see that the statin patients were slower (or
never) to progress to severe disease, and faster to recover. More quantitatively, they
computed the usual hazard ratio and associated logrank $p$-value, to assess whether the curves are
statistically significantly different, not just visually. From table 2 in the paper:
- For time to recovery, statin use was significant ($p \leq 0.004$), but ACE/ARB use was not.
- For time to progression to severe disease, nobody made the cut. (Though the authors, as always being hopeful, thought they could see a weak trend. Crusty old statisticians like your Weekend Editor are seldom sympathetic to this.)
- Age cohort (10 year groups) was also statistically wildly significant, in the bad direction ($p \leq 0.001$). (There are, alas, as yet few treatments for aging. Perhaps Aubrey de Grey will one day have something to say about that?)
There’s more, but that’s kind of the bottom line:
- If you’re taking a cholesterol-lowering drug, particularly a statin, congratulations on your partial defense against severe COVID-19. Keep doing that, because your doctor has your best interests at heart here. [4]
- If you’re taking an ACE/ARB inhibitor for blood pressure, it doesn’t help against COVID-19, but it also doesn’t hurt, so congratulations on treating your high blood pressure. Keep doing that, again following your doctor’s advice.
Molecular mechanism of action of statins & cholesterol on cell entry by SARS-COV-2
 Daniels et al. then speculate about all kinds of mechanisms, but they’re all over the map and show
no data. Fortunately, the next paper (Wang, et al., reference 3) does exactly that.
Daniels et al. then speculate about all kinds of mechanisms, but they’re all over the map and show
no data. Fortunately, the next paper (Wang, et al., reference 3) does exactly that.
They explore a model which is at once pretty simple (A causes B causes C causes D…) and laughably complex, as things always seem to be in biology where the Law of Unintended Consequences runs riot. They are very careful to do a number of “rescue assays” to see if interrupting the chain at various points has the effect one would predict.
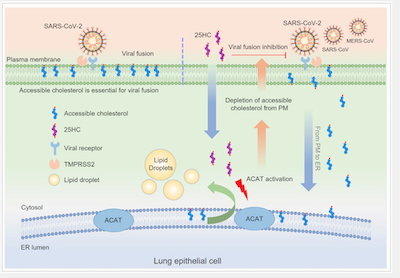 The basics: viruses stimulate the innate immune system,
which causes the release of interferons of various kinds,
which turns on a gene called CH25H, which makes
something called 25-hydroxycholesterol (25HC),
which activates another gene called ACAT on the ER
membrane (and elsewhere), which then internalizes
cholesterol usually found in the cell membrane. This low-cholesterol membrane then
prevents the virus from being endocytosed, or absorbed into the cell. It stays outside,
where it can’t do much damage and where it can be recognized by the adaptive immune system
(eventually). So… A causes B causes C causes… for 6 steps!
The basics: viruses stimulate the innate immune system,
which causes the release of interferons of various kinds,
which turns on a gene called CH25H, which makes
something called 25-hydroxycholesterol (25HC),
which activates another gene called ACAT on the ER
membrane (and elsewhere), which then internalizes
cholesterol usually found in the cell membrane. This low-cholesterol membrane then
prevents the virus from being endocytosed, or absorbed into the cell. It stays outside,
where it can’t do much damage and where it can be recognized by the adaptive immune system
(eventually). So… A causes B causes C causes… for 6 steps!
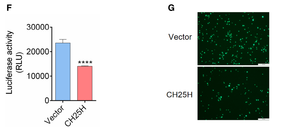 They present evidence that cells infected with SARS-COV-2 do indeed raise their expression
of CH25H, and that this is also observed in patient samples. They then showed that when
they forced overexpression of CH25H in cell lines, SARS-COV-2 entry to cells was lowered,
and that was statistically significant by $t$-test. (Showing Figure 1 C & D here: green
spots show cells infected by SARS-COV-2, while the bar chart shows the data after image
quantitation. It is significant: $p \leq 0.0001$ by $t$-test.)
They present evidence that cells infected with SARS-COV-2 do indeed raise their expression
of CH25H, and that this is also observed in patient samples. They then showed that when
they forced overexpression of CH25H in cell lines, SARS-COV-2 entry to cells was lowered,
and that was statistically significant by $t$-test. (Showing Figure 1 C & D here: green
spots show cells infected by SARS-COV-2, while the bar chart shows the data after image
quantitation. It is significant: $p \leq 0.0001$ by $t$-test.)
Then, the objections start:
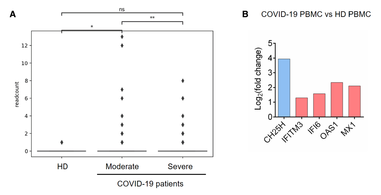 But is this just a cell line thing, of no relevance to actual patients? They also did
something similar in patient samples (Figure EV-1), to similar effect. On the left are
the gene expressions levels of CH25H in epithelia from bronchoalveolar lavage (basically
they wasn your lungs from the inside and collect the goop; doesn’t that sound pleasant?)
in healthy, moderate, and severe COVID patients. The differences are statistically
significant by $t$-test ($p \leq 0.01$ or $p \leq 0.05$). On the right are the gene
expression fold inductions (ratio of infected to normal patient levels); note that CH25H
is induced about 4 times more in infected patients. (They did not report significance
here, but it looks good and we could follow their reference chain to find the original
data and check, should that become an issue.) So it’s not just a cell line thing.
But is this just a cell line thing, of no relevance to actual patients? They also did
something similar in patient samples (Figure EV-1), to similar effect. On the left are
the gene expressions levels of CH25H in epithelia from bronchoalveolar lavage (basically
they wasn your lungs from the inside and collect the goop; doesn’t that sound pleasant?)
in healthy, moderate, and severe COVID patients. The differences are statistically
significant by $t$-test ($p \leq 0.01$ or $p \leq 0.05$). On the right are the gene
expression fold inductions (ratio of infected to normal patient levels); note that CH25H
is induced about 4 times more in infected patients. (They did not report significance
here, but it looks good and we could follow their reference chain to find the original
data and check, should that become an issue.) So it’s not just a cell line thing.
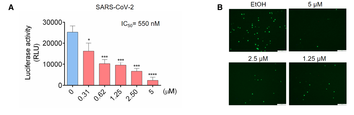 But is this dependent on the dose of 25HC, as you would expect? Yes. In Figure 2 A&B
shown here, we see that both quantitatively and visually with a GFP fluorescence image.
More 25HC leads to less SARS-COV-2 entry into cells.
But is this dependent on the dose of 25HC, as you would expect? Yes. In Figure 2 A&B
shown here, we see that both quantitatively and visually with a GFP fluorescence image.
More 25HC leads to less SARS-COV-2 entry into cells.
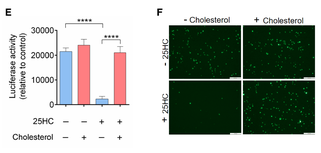 But can you “rescue” the effect (get the virus to work again) by adding lots of
cholesterol? Maybe you can overload things, so CH25H/25HC/ACAT and company can’t deplete
the membrane cholesterol any more. Figure 3 shows this, by doing all combinations of
adding 25HC or not and adding cholesterol or not. All the expected differences are
significant, by $t$-test with $p$ in the range of 0.01 to 0.0001.
But can you “rescue” the effect (get the virus to work again) by adding lots of
cholesterol? Maybe you can overload things, so CH25H/25HC/ACAT and company can’t deplete
the membrane cholesterol any more. Figure 3 shows this, by doing all combinations of
adding 25HC or not and adding cholesterol or not. All the expected differences are
significant, by $t$-test with $p$ in the range of 0.01 to 0.0001.
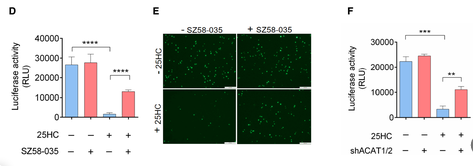 But is ACAT really the gene involved in this mechanism? If you “knock down” the ACAT
gene with shRNA or an experimental ACAT inhibitor compound, you’d expect the virus to be
able to get in again, i.e., another rescue experiment. Figure 4 shows this. Yes: whether
by shRNA to prevent ACAT from
being made, or with a molecule to inhibit ACAT activity, once ACAT is shut down the effect
goes away.
But is ACAT really the gene involved in this mechanism? If you “knock down” the ACAT
gene with shRNA or an experimental ACAT inhibitor compound, you’d expect the virus to be
able to get in again, i.e., another rescue experiment. Figure 4 shows this. Yes: whether
by shRNA to prevent ACAT from
being made, or with a molecule to inhibit ACAT activity, once ACAT is shut down the effect
goes away.
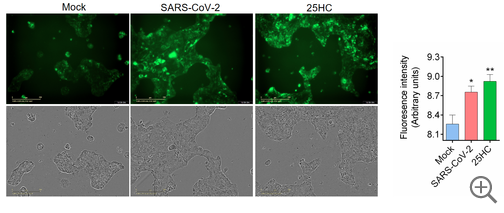 But does SARS-COV-2 itself actually activate ACAT and 25HC? Figure EV4 shows that the
answer is yes. So basically, these data taken together shows that SARS-COV-2 activates
CH25H, then 25HC is made, then ACAT is activated, then cholesterol is pulled in from the
cell membrane, then virus entry is blocked.
But does SARS-COV-2 itself actually activate ACAT and 25HC? Figure EV4 shows that the
answer is yes. So basically, these data taken together shows that SARS-COV-2 activates
CH25H, then 25HC is made, then ACAT is activated, then cholesterol is pulled in from the
cell membrane, then virus entry is blocked.
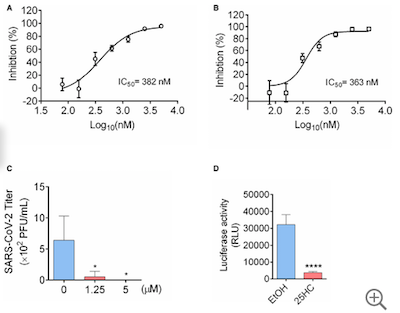 But does that work in something like a human lung, not just in cell cultures? Figure
5 shows that the answer is yes. They grew lung organoids (basically a cell culture in
something like soft agar where lung cells can build 3d structures that are at least
somewhat similar to actual lung tissue). The dose response curves show that the
inhibition of virus by 25HC is still dose-dependent in cell lines, and that the potency
looks to be around 0.5$\mu$M. Also, the bar charts below show a dramatic effect on virus
blocking in lung organoids. So it’s both dose-dependent, and works in something like an
organ.
But does that work in something like a human lung, not just in cell cultures? Figure
5 shows that the answer is yes. They grew lung organoids (basically a cell culture in
something like soft agar where lung cells can build 3d structures that are at least
somewhat similar to actual lung tissue). The dose response curves show that the
inhibition of virus by 25HC is still dose-dependent in cell lines, and that the potency
looks to be around 0.5$\mu$M. Also, the bar charts below show a dramatic effect on virus
blocking in lung organoids. So it’s both dose-dependent, and works in something like an
organ.
There’s a lot more there, but it’s pretty convincing at this point. The only thing you might wonder is why bother with statins as a back-door path to all this, when you could administer 25HC itself and get the effect more directly? Reference 5 points out exactly this. But… practically speaking, statins are an old and very safe drug taken by millions with excellent effect on cardiac health, while 25HC use as a drug is a totally new thing, requiring new trials. Still, it’s worth investigating, I suppose.
Summary
Ok, so that was a long trip. I think the basics are:
- We have good clinical evidence in actual patients that statins are a good thing. This is definitely true for progression to severe disease and for recovery time, though not so much for death rates.
- We have very good, detailed evidence of the pathway by which this happens in cells and in human-like organoids: virus → innate immune response → interferons → CH25H → 25-hydroxy-cholesterol → ACAT activation → membrane depletion of cholesterol → reduced virus entry.
- We have long clinical history that taking statins also reduces cholesterol, at least partly in cell membranes. (Usually it’s measured in blood sera, not cell membranes.)
- There may be better ways to do this (direct administration of 25-hydroxycholesterol, perhaps directly to the lungs with a nebulizer?), but statins seem to help.
- Statins, unlike remdesivir, are long-approved, well-understood, and cheap.
- A few missing items: expansion beyond a single-center trial (replication is important!), and a link between clinically observed response in patients and the specific mechanism involving interferon/CH25H/25HC/ACAT/membrane cholesterol depletion vs almost any other mechanism, and finally a response biomarker telling us which COVID-19 patients are most likely to benefit to benefit from statin therapy.
So take your statins, if you doctor has prescribed them!
Notes
1: In the interests of disclosure, one should note that both your Weekend Editor and Weekend Editrix take a statin and an ACE inhibitor. So, if anything, we want something like this to be true, as it would grant us some extra protection. And my prior says that ACE2 (binding site of SARS-COV-2) and ACE (the blood pressure med target) are homologous proteins, so maybe inhibiting ACE has some crosstalk to ACE2. Be appropriately skeptical of my prejudices. (Comme d’habitude!)↩
2: Also, your humble Weekend Editor once worked for a company which did some of the first gene sequencing around ACE2, and thus had an early ACE2 inhibitor around 2001. It failed clinical trials in 2 separate disease areas, and was later out-licensed. Nothing much came of it, though now its interest in blocking the COVID-19 entry point has returned. Experiencing 2 clinical trial failures would make me even more skeptical in this area. ↩
3: For example, age is a risk factor for severe COVID. But it’s also a risk factor for needing blood pressure and high cholesterol meds. So if you didn’t account for age as a confounding covariate, you might conclude that older people take cholesterol meds, and older people get severe COVID-19, so maybe the cholesterol med caused it — when the opposite is true, it protects. Intuitively, you first regress the log odds ratio of severe disease on all the confounders (age, sex, obesity, …) and then do prediction using the cholesterol/bp meds on the residuals. (In fact it happens slightly differently, but that’s often the way to explain it for the first time and have people more or less get it.)↩
4: And most especially: please don’t ever take anything you read here as medical advice! Your humble Weekend Editor is not an MD, but a totally different sort of doctor (PhD). The closest we’ll come to medical advice is: (a) get a doctor, and (b) do what your doctor says. Right-wing political mischief with health insurance in the US is a problem everybody here has, so do your best with that. ↩
References
-
H Buschman and J Vazquez, “Statins reduce COVID-19 severity, likely by removing cholesterol that virus uses to infect.”, UC San Diego News Center, 2020-Sep-23. Also reported in Science Daily.
-
L Daniels, et al., “Relation of Statin Use Prior to Admission to Severity and Recovery Among COVID-19 Inpatients”, AmJCardiol, 2020-Sep-15 DOI: 10.1016/j.amjcard.2020.09.012.
-
S Wang, et al., “Cholesterol 25‐Hydroxylase inhibits SARS‐CoV‐2 and coronaviruses by depleting membrane cholesterol”, EMBO Jnl, e106057, 2020-Oct-05. DOI: 10.15252/embj.2020106057.
-
L Fang, et al., “Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection?”, Lancet Resp Med, 8:4, 2020-Apr-01. DOI: 10.1016/S2213-2600(20)30116-8.
-
S Zu, et al., “25-Hydroxycholesterol is a potent SARS-CoV-2 inhibitor”, Nature Cell Res, 2020-Aug-18.

Gestae Commentaria
Comments for this post are closed pending repair of the comment system, but the Email/Twitter/Mastodon icons at page-top always work.